Chapter 13: Cannabinoids
2nd edition as of August 2022
Chapter Overview
For this chapter, we will discuss a class of drugs that shares many similarities to opioids. Both opioids and cannabinoids are drugs that are extracted from plants with a long history of use, both act on endogenous receptor systems named after the exogenous drugs, and both have largely inhibitory effects on neural activity. Both have also been under intense scrutiny in recent decades, although for different reasons. Whereas opioids have invaded public awareness through the opioid epidemic, most people are probably familiar with cannabis, or marijuana, because of intense current efforts to legalize the drug in the U.S. In this chapter, we will examine the key substances in cannabis and learn about where the effects of marijuana come from, as well as how its medical and recreational use has been changing in recent years.
Chapter Outline
13.1. Cannabinoids and Overview
- 13.1.1. The Cannabis Plant
- 13.1.2. History and Legislation
- 13.1.3. The Endocannabinoid System
- 13.2.1. Administration and Pharmacokinetics
- 13.2.2. Mechanisms of Action and Effects
- 13.2.3. Tolerance, Withdrawal, and Long-Term Effects
- 13.2.4. Medical Marijuana and Cannabinoid Medications
Chapter Learning Outcomes
- Describe and contrast cannabinoids, hemp, and marijuana and outline the history of cannabis in relation to legislation.
- Describe the endocannabinoid system.
- Describe the pharmacology of cannabis, to include its administration, pharmacokinetics, mechanism of action and effects, tolerance, withdrawal, and long-term effects.
13.1. Cannabinoids Overview
Section Learning Objectives
- Describe the cannabis plant and identify its main active ingredients.
- Differentiate between hemp and marijuana.
- Compare and contrast Δ9-tetrahydrocannabinol (THC) and cannabidiol (CBD).
- Describe the history of marijuana.
- Explain the endocannabinoid system and define its primary receptors and transmitters.
A cannabinoid is a biologically active compound found in cannabis. There are over 100 such substances that can be isolated from cannabis, only some of which are psychoactive. In this chapter, we will focus on two of the most common cannabinoids and compare the effects of cannabis containing them in different amounts.
13.1.1. The Cannabis Plant
All cannabinoids originate from the resin produced by the Cannabis genus of plants, which is often differentiated into three variations. The following table is a comparison of the three most common species: Cannabis sativa, Cannabis indica, and Cannabis ruderalis.
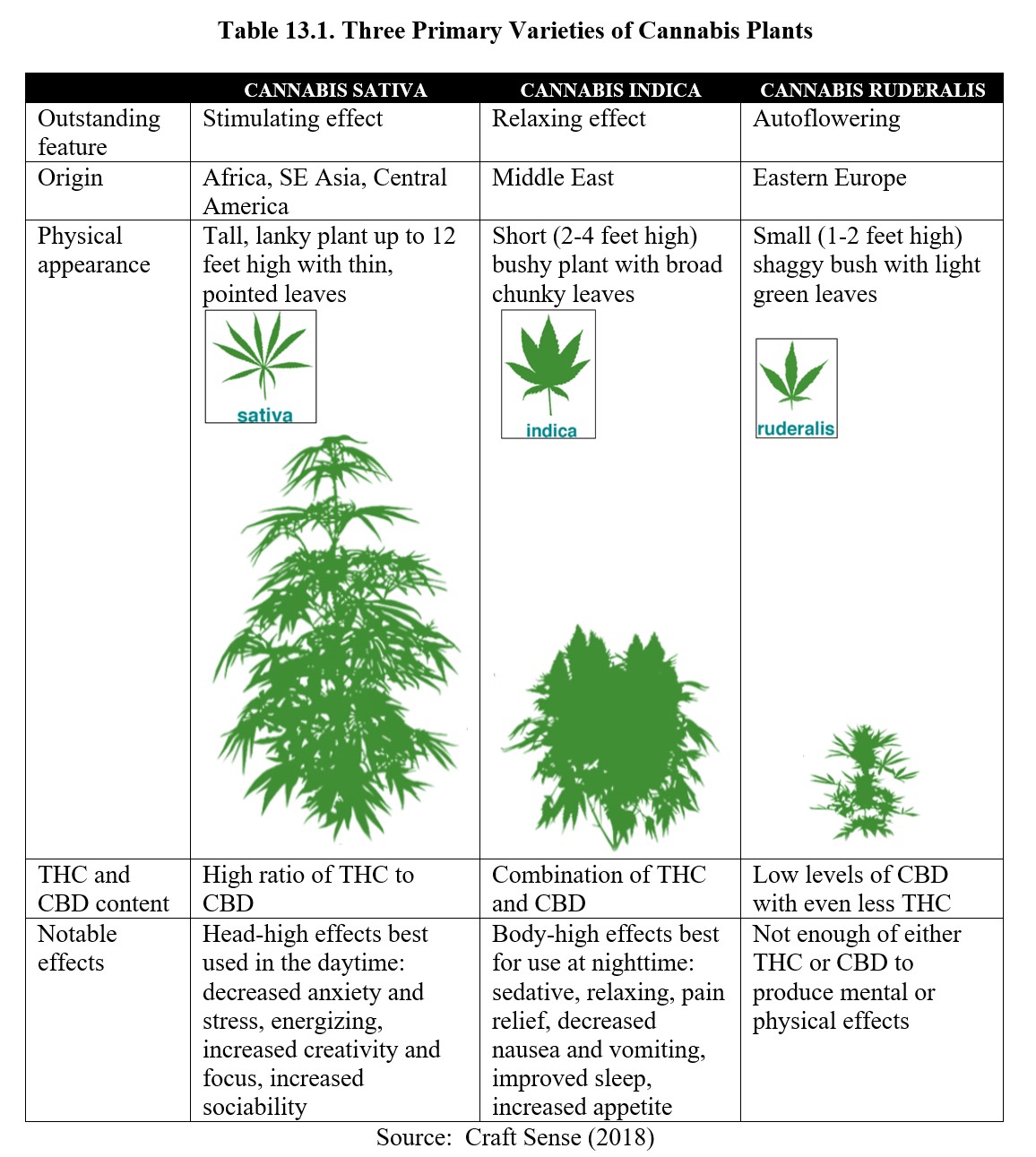
Although these variations are often presented as distinct species, in reality, there is a large amount of overlap. Most strains of cannabis are hybrids that have undergone crossbreeding to enhance particular properties (i.e., to produce a stronger high, or to be easier to grow indoors). For example, Cannabis ruderalis is notable for being autoflowering, meaning that it will flower according to age rather than light stimulus and takes a shorter time before harvesting. Cannabis sativa and Cannabis indica are photoperiod flowering, dependent on the light cycle, and take a longer time before harvesting. Despite its low THC content, Cannabis ruderalis is frequently crossbred with other strains to transfer the autoflowering trait and increase the yield in the hybrid. As a result, it is often more useful to distinguish between different strains by examining the type and concentration of cannabinoids each contains.
The most notable cannabinoid is delta-9-tetrahydrocannabinol, also referred to as Δ9-THC or simply THC. (Despite there being other tetrahydrocannabinols, when referring to THC in this chapter, we will be specifically talking about Δ9-THC). This is the main psychoactive component found in cannabis and is typically sought after for recreational use.
Another well-known cannabinoid is cannabidiol or CBD. Unlike THC, CBD is non-psychoactive. Although it can still interact with the nervous system, it does not produce psychoactive effects. Its presence may counteract some of the effects of THC (more on that later). Because of this, CBD is sought more often for therapeutic purposes.
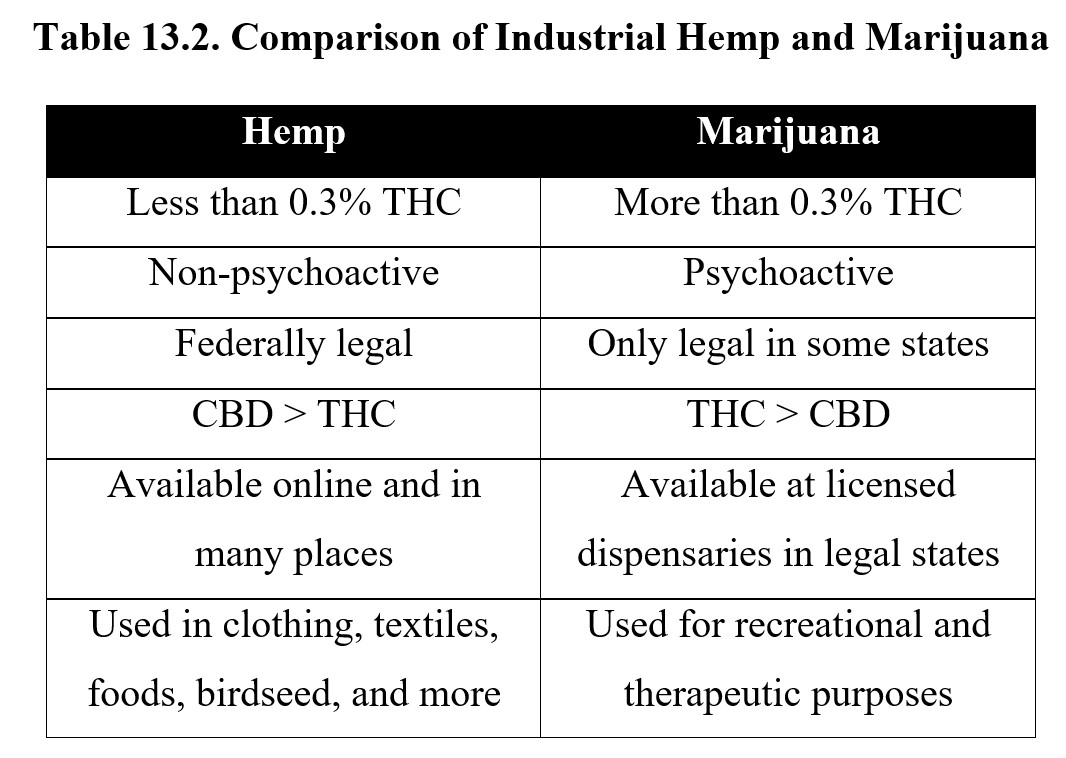
As THC is the main psychoactive cannabinoid, another way of classifying cannabis strains is by THC content. Cannabis with low THC content (less than 0.3%) is also called hemp or industrial hemp. It has little THC and is non-psychoactive. It can be harvested for its fiber, seeds, and CBD but is never used recreationally. Hemp fibers have many industrial uses such as the manufacturer of clothing, textiles, and rope. Hemp can also be processed into foods, birdseed, and even biofuel. Legally, if the THC content is higher than 0.3%, it is then called marijuana. Because it is psychoactive, marijuana is only legal in some states and is used primarily for its psychoactive effects. See the table below for a comparison of the two.
The THC content of marijuana is not fixed and has increased in recent decades. In the 1970s, the average THC content was 1–4%, but this has risen to 10–15% in recent years, with some strains containing as high as 30% THC content. This increase in potency is caused by hybridization and also changes in growing methods that selectively breed strains with a higher THC content that produce more resin.
One final thing worth noting is that it is possible to artificially manufacture substances that produce similar effects as cannabinoids. These compounds are known as synthetic cannabinoids and can have significantly different effects compared to cannabis. To distinguish the two types, cannabinoids extracted from plants are called phytocannabinoids (from phuton in Greek for “plant”). Synthetic cannabinoids will be covered in a later chapter on new drugs of abuse.
13.1.2. History and Legislation
The history of cannabis is much like the history of opium poppies and coca leaves. The earliest written record of cannabis use occurred in 2700 B.C. when Chinese emperor Shen Nung included it in a book of medicinal herbs. Cannabis originated in central Asia and India, but its cultivation and use spread throughout the world. In 200 B.C., cannabis was used in ancient Greece as a remedy for earaches, edema, and inflammation. From 800–1500 A.D., cannabis was used as medicine in the Arabian Peninsula, where it was also used as hashish for its mood-changing properties.
The history of cannabis in the U.S. dates back to the 17th century, where it was grown in the colonies for hemp products. British colonies were required by law to grow hemp for producing cordage, sails, and cloth for the Royal Navy and mercantile marine. Marijuana saw its popular use as a remedy in the 1800s and early 1900s and was an ingredient in many medicines and tinctures at the time. Marijuana was also commonly smoked for recreational purposes at the time.
The legal status of marijuana changed in 1937 with the passage of the Marihuana Tax Act. The new law required registration and a tax levy on the importation and commercial use of marijuana. This effectively outlawed the non-medical or recreational use of marijuana. It also legitimized the term marijuana to refer to cannabis products, as it was previously only a slang term for cannabis. Because of the tax imposed by the act, medical use of marijuana also declined.
In 1970, the Controlled Substances Act classified marijuana as a Schedule I drug with no accepted medical use, meaning it was illegal to prescribe it for medical treatment. The initial placement of the drug in Schedule I was intended to be provisional because of the lack of studies on its safety. But in 1972, the Nixon administration refused to reschedule marijuana despite the report of a national commission recommending its decriminalization.
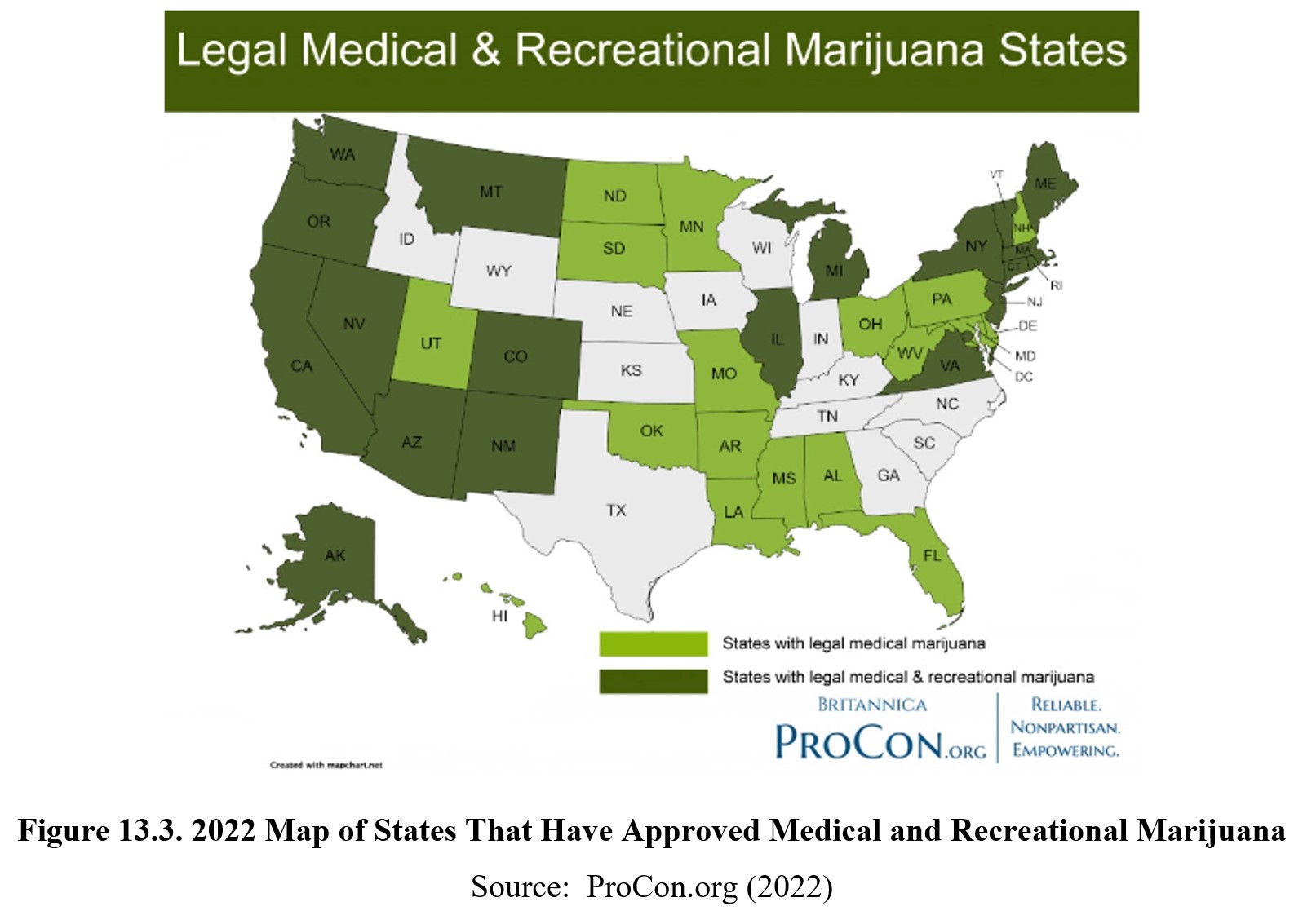
The Schedule I status of marijuana has been repeatedly challenged in subsequent decades. To this date, it is still federally classified as an illegal drug. Despite this, some exceptions have been carved out over the years, and state legislatures have passed laws legalizing its use. California was the first state to legalize medical cannabis in 1996, while Washington and Colorado were the first to legalize recreational use in 2012. Since then, 37 states and the District of Columbia have legalized medical marijuana. Canada also legalized marijuana in 2018.
Up until 2018, there was no distinction between industrial hemp and hemp grown for marijuana. The Hemp Farming Act passed by the U.S. Congress formally differentiated between hemp and marijuana using the THC content threshold of 0.3% mentioned above, effectively legalizing certain cannabinoids at the federal level for the first time. Hemp with greater than 0.3% THC was defined as marijuana, and hemp with less than 0.3% THC was considered industrial and categorized as an agricultural commodity.
13.1.3. The Endocannabinoid System
Similar to opioids, the effects of cannabis were well-established long before we knew how and why cannabis was capable of interacting with the brain. The eventual discovery of an endogenous system of cannabinoid receptors and ligands referred to as the endocannabinoid system, did much to further our understanding of how cannabis achieves its effects.
The Endocannabinoid System [4:13]
Two types of cannabinoid receptors have been identified: the cannabinoid receptor type 1 (CB1) and cannabinoid receptor type 2 (CB2). Both are G-protein coupled receptors. CB1 receptors are found throughout the CNS and peripheral tissues while CB2 receptors are largely located outside of the nervous system on immune cells. The pharmacological effects of cannabinoids are mainly determined by their affinity and efficacy for CB1 receptors, although there are likely to be additional types of cannabinoid receptors not yet identified that account for other effects of cannabinoids.
Endogenous ligands for these receptors are called endocannabinoids as opposed to the exogenous phytocannabinoids found in plants. The first endocannabinoid discovered was named N-arachidonylethanolamine (AEA) or anandamide after the Sanskrit word Ananda, meaning joy or bliss. The other primary endocannabinoid is 2-arachidonoylglycerol (2-AG). Anandamide is an agonist of the CB1 receptor, while 2-AG is an agonist of the CB2 receptor, although the two have similar chemical structures and can interact with both receptor types. There are an additional one hundred cannabinoid-related substances found in the cannabis plant, but these have not been studied as intensively as anandamide and 2-AG.
Anandamide and 2-AG are not synthesized and stored in synaptic vesicles. Rather, both are made on-demand and released almost immediately into the synaptic space. They tend to have short half-lives and are quickly metabolized by enzymes. Anandamide is metabolized by fatty acid hydrolase (FAAH) into arachidonic acid, while 2-AG is metabolized by monoacylglycerol lipase (MAG-L) into fatty acids and glycerol.
Endocannabinoids act as retrograde neurotransmitters at many synapses. Instead of being released by the presynaptic neuron, endocannabinoids are released by the postsynaptic neuron, whereupon they travel across the synapse in the opposite direction from most transmitters and bind to CB receptors on the presynaptic neuron (see image below). This reduces neurotransmitter release, causing an inhibitory effect.
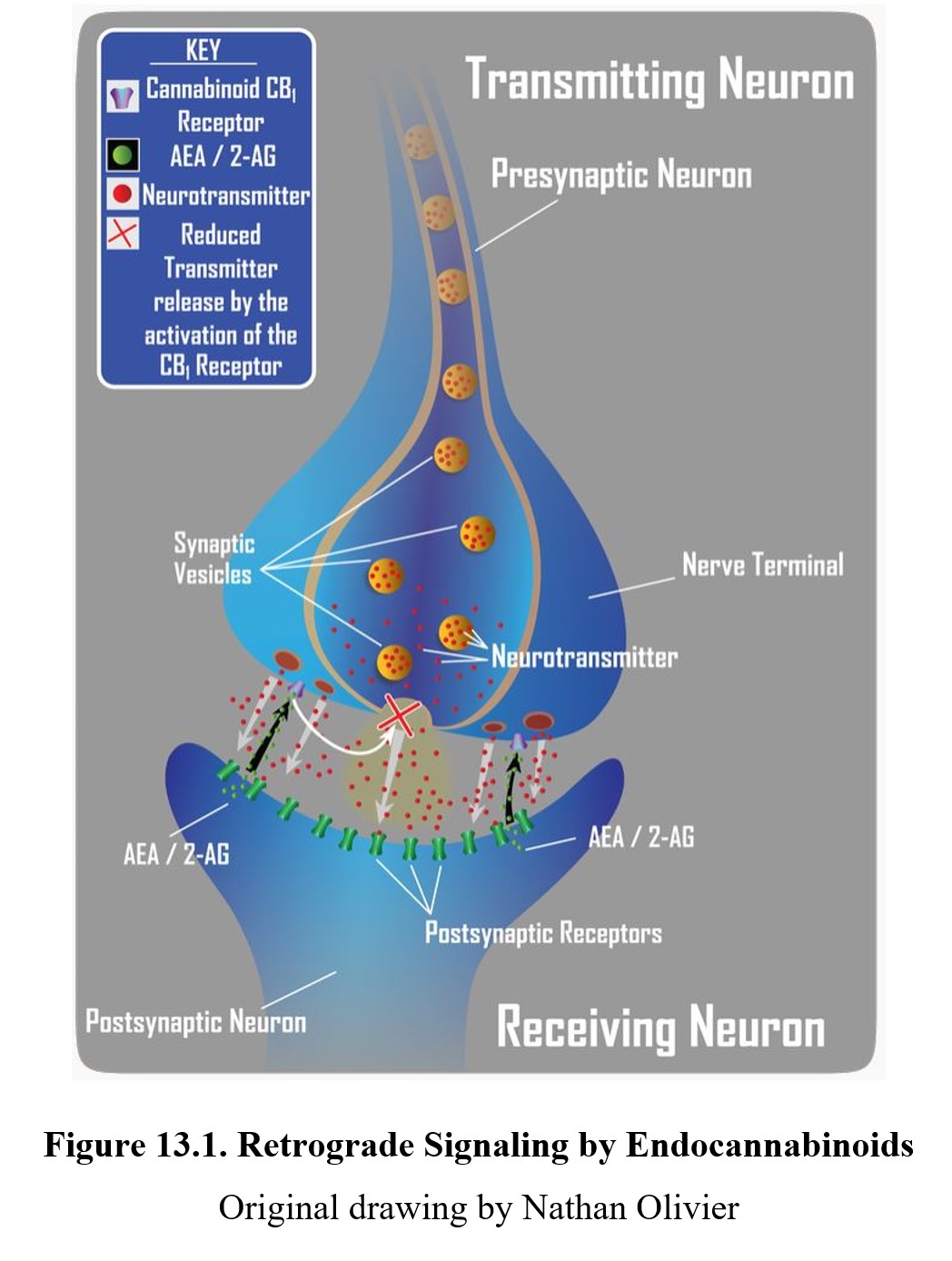
An example of the role of retrograde transmission in the modulation of a physiological function is in nociception and antinociception. In the example above, excessive neurotransmission across a Substance P or glutamate synapse in the pain pathway is responsible for the perception of intense pain. When overly stimulated, the postsynaptic neuron produces and releases endocannabioid (anandamide or 2-AG) which travels from the postsynaptic neuron back to the presynaptic neurons. Activation of cannabinoid CB1 receptors reduces the amount of primary transmitter released into the synapse. This reduces the intensity of the pain signal and may underlie the analgesic effect of cannabinoids. Endocannabinoids can also produce excitatory effects such as stimulation of appetite. This likely occurs through endocannabinoids reducing the inhibitory effects of GABA neurons.
As such, the endocannabinoid system is an important method of neuromodulation. Although our understanding of the endocannabinoid system is still evolving, preliminary research suggests that the system is involved in many functions, such as regulating appetite, sleep, mood, and pain. Scientists believe that the main role of the endocannabinoid system is the maintenance of homeostasis. The endocannabinoid system also seems to play an important role in forgetting by suppressing long-term potentiation in the hippocampus.
13.2. Pharmacology
Section Learning Objectives
- Describe the pharmacokinetics of marijuana.
- Describe the physiological and behavioral effects of marijuana.
- Discuss tolerance and dependence on marijuana.
- Describe the long-term adverse and toxic effects of marijuana.
- Define medical marijuana and explain the use and origin of Charlotte’s Web.
- Describe the approved cannabinoid drugs and their clinical uses.
As mentioned in the previous section, cannabinoids are many biologically active substances found in cannabis. Because there are numerous cannabinoids with varying properties, in this section, we will zero in on THC and CBD in particular. When talking about the psychoactive effects of cannabis or marijuana, remember that this is due to THC, its psychoactive component, and not due to CBD, which is non-psychoactive.
13.2.1. Administration and Pharmacokinetics
Cannabis is most often inhaled or ingested. Cannabis can be rolled into a cigarette or joint, or it can be smoked with the aid of hookahs or bongs. Second-hand exposure to the smoke can result in a contact high. Oral administration occurs when eating hash oil or preparations that contain cannabis, such as brownies.
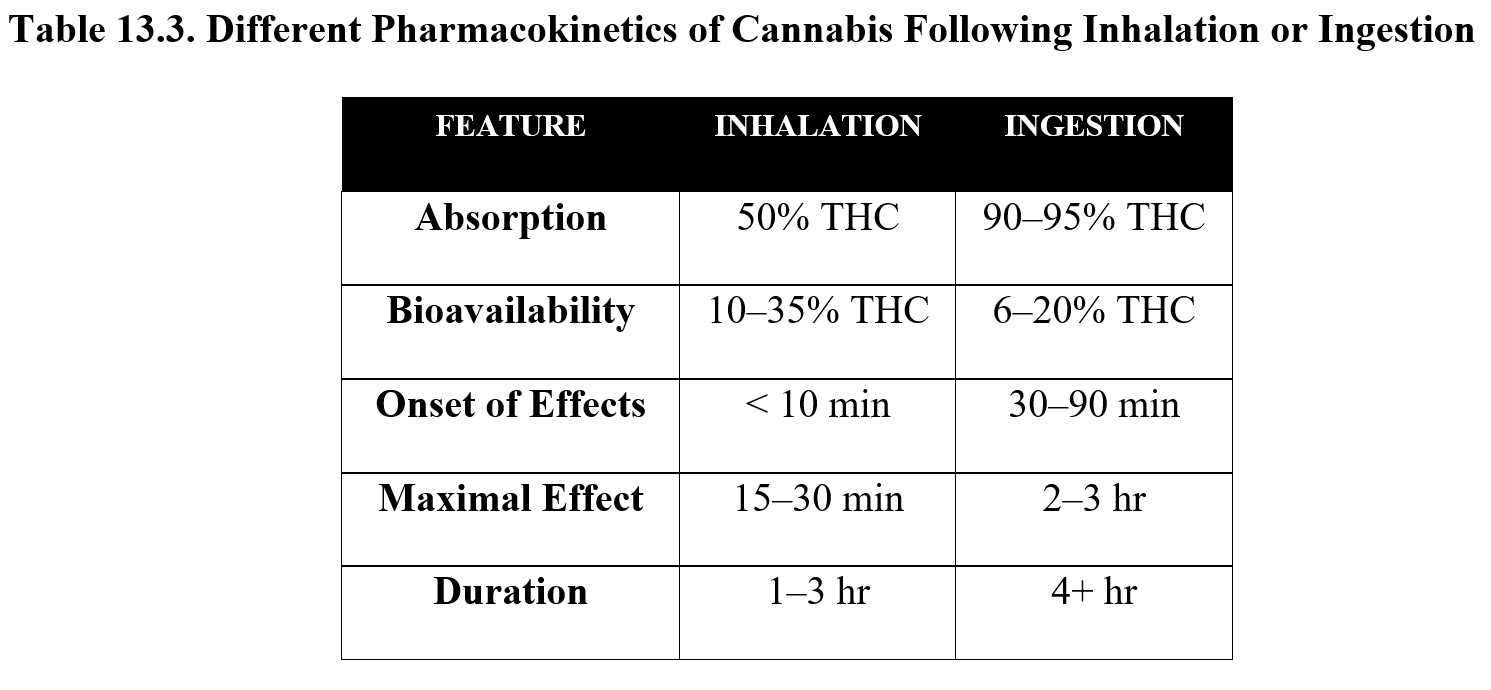
When smoked, about 50% of the THC content is absorbed, with the remainder being released into the environment. Once absorbed into the bloodstream by the lungs, peak concentrations of THC are achieved within minutes, with maximal effects occurring within half an hour. The duration of the effects usually lasts for 1–3 hours depending on the amount administered.
In comparison, oral ingestion results in a higher rate of absorption of THC (about 90–95%), but due to first-pass metabolism in the liver, its bioavailability drops to around 6–20%. In addition, the effects are much more delayed compared to smoking. The onset of action occurs after 30–90 minutes, while peak effects may not occur until 2–3 hours after administration. Duration is prolonged, with effects lasting for at least 4 hours. Compare the two routes below:
Because it is lipid-soluble, THC can cross the blood-brain barrier and is readily distributed throughout body tissue. Metabolism of THC occurs primarily in the liver through cytochrome P450 enzymes, in particular CYP2C9, which converts Δ9-THC into 11-OH-Δ9-THC (often referred to simply as 11-hydroxy-THC). An active metabolite itself, 11-hydroxy-THC has similar effects to THC and is, in turn, metabolized into glucuronide conjugates and other inactive metabolites before being excreted.
THC tends to have a half-life of around 20–50 hours, although it can be detected for days or up to a month after administration. This is in part because it can accumulate in fat cells and is gradually re-released into the bloodstream over time. Most of the drug (more than 65%) is excreted in the feces, with about 20% excreted through the urine.
13.2.2. Mechanism of Action and Effects
The effects of cannabinoids are largely mediated by the CB1 receptor. THC is a partial agonist with a high affinity for the CB1 receptor, similar to the endocannabinoid anandamide. Some synthetic cannabinoids have a much higher affinity for the CB1 receptor compared to anandamide and THC. As mentioned previously, there may be additional cannabinoid receptors responsible for some of the effects of THC that are being investigated or yet to be discovered.
CBD is unique in that it may counteract the effects of THC. This is because CBD is a negative allosteric modulator of the CB1 receptor. In other words, when CBD binds to the CB1 receptor, it changes the conformation of the receptor site so that THC cannot bind to it as easily. At the same time, CBD also interacts with the receptors for other neurotransmitters, such as serotonin, and may enhance the effects of THC in some cases.
Because the endocannabinoid system is found throughout the CNS and peripheral tissues, administration of THC produces a variety of effects. Perhaps the most well-known physiological effect is an increase in appetite, also known as the munchies. THC also increases heart rate and blood pressure, reduces motor activity, and relaxes the airways. It also dilates the blood vessels in the conjunctiva, the membrane that covers the front surface of the eye. The presence of bloodshot eyes is a clear sign of cannabis use. THC can also cause drowsiness, dizziness, dry mouth and throat, and reduce restful sleep by suppressing REM sleep.
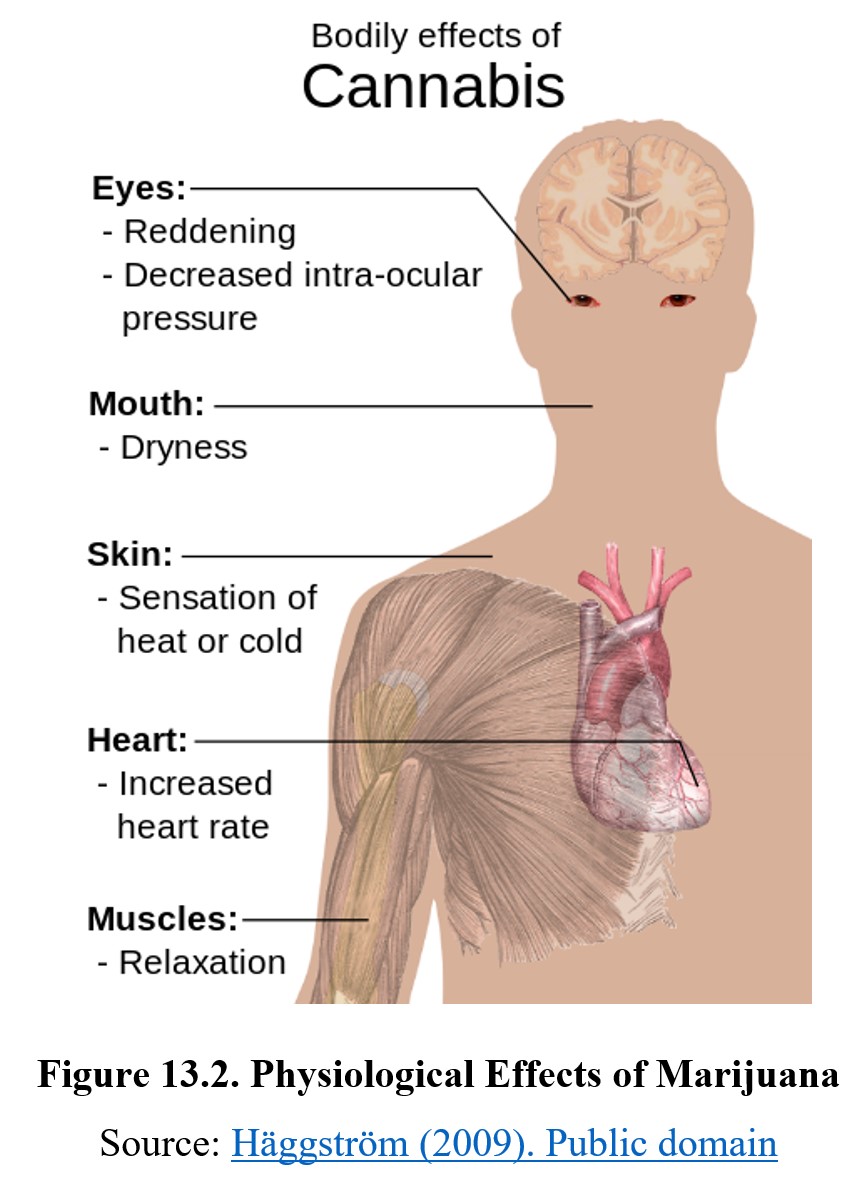
Cannabis also has effects on the endocrine system and the release of sex hormones. In men, the release of testosterone is decreased, which can result in erectile dysfunction and reduced sperm count and motility. In women, a decrease in progesterone can result in reduced sexual pleasure, disruption of menstrual cycles, reduced fertility, and an increased risk of premature birth.
The subjective effects of marijuana are dose-dependent and generally follow three stages. The buzz occurs at low doses and is characterized by feelings of dizziness, light-headedness, and tingling sensations. This is usually followed by the high, which may entail euphoria, exhilaration, and disinhibition. In first-time users or at very high doses, this may also result in anxiety, paranoia, and panic attacks. The effects on mood may be influenced by the setting, with pleasant settings causing positive effects and unpleasant settings causing negative effects. Finally, the stoned phase involves relaxation and altered perception. Users often report enhanced senses and the feeling that time passes more slowly.
Marijuana also interferes with judgment and short-term memory, which can increase risky behaviors and make it difficult to retain information. High doses of marijuana can cause paranoia and psychosis. Marijuana causes dose-dependent impairment of psychomotor activity, which results in decreased alertness, concentration, coordination, and reaction time. At low doses, there is relatively little effect, but the impairment increases with larger doses. In addition, combining marijuana with alcohol produces an additive effect, which can cause severe impairment and increase the risk of a car accident.
The impact of smoking marijuana is similar to the impact of smoking cigarettes. Smoking joints can cause inflammation in the lungs and can lead to chronic obstructive pulmonary disease (COPD), emphysema, and chronic bronchitis. Marijuana cigarettes reportedly contain more tar than tobacco cigarettes, and cannabis tar contains greater amounts of carcinogens. However, the risk of lung cancer is less than cigarettes. Marijuana smokers are at greater risk of pulmonary complications because they tend to inhale deeply and hold the smoke in their lungs longer. Secondhand smoke from marijuana can produce a contact high as well as serious breathing issues, especially in children exposed to marijuana smoke in an enclosed environment,
13.2.3. Tolerance, Withdrawal, and Long-Term Effects
Regular use of cannabis can cause tolerance to the behavioral and subjective effects of cannabis. If used frequently, withdrawal can occur upon stopping. Compared to other drugs we have examined, cannabis withdrawal is usually mild and rarely life-threatening. Symptoms include craving, irritability, anxiety, lack of appetite, sleep disturbance, and depression. Some users may also experience physical symptoms such as tremors, sweating, headache, stomach pain, and fever. Withdrawal tends to last for up to two weeks, with peak symptoms occurring 2–6 days after the cessation.
Activation of CB1 receptors causes an increase in dopamine release in the nucleus accumbens, although the exact mechanism through which this occurs is poorly understood at the time. This increase in dopamine is responsible for some of the euphoric effects of cannabis. Although dependence is possible, it is not common, and some researchers believe that dependence is psychological, rather than physiological. Tolerance and dependence are more likely to develop with frequent use of strains that are high in THC content.
Although dependence and addiction are uncommon, they do occur. The DSM-V describes cannabis use disorder as a type of substance use disorder and lists 11 criteria to evaluate its severity. Similar to other substance use disorders, cannabis use disorder is characterized by cravings, preoccupation with obtaining cannabis, compulsive use despite negative consequences, and relapse after stopping use.
Frequent or heavy use of cannabis can cause many negative long-term effects, especially if the use begins in adolescence. Addiction occurs in 9% of overall users. This number increases to 17% in those who began in adolescence and 25–50% of those who use cannabis daily. Early use is also associated with changes to brain development and cognitive impairment, as well as diminished life satisfaction and educational achievement.
Chronic smoking of cannabis also increases the risks of certain respiratory diseases, such as emphysema and bronchitis. But cannabis use is not as strongly associated with lung cancer because some compounds in marijuana may suppress tumor growth. Smoking cannabis can also increase cardiovascular problems if there is a preexisting heart condition.
13.2.4. Medical Marijuana and Cannabinoid Medications
In 1996, California was the first state to approve medical marijuana as a voter initiative. In 2012, voters in Colorado and Washington states approved ballot initiatives to legalize recreational marijuana. By May 2022, 38 states and the District of Columbia were the first states have legally recognized the medical and recreational use of marijuana. The landscape of cannabis continues to change.
Although marijuana is most well-known for being a recreational drug, it is also sometimes used to treat diseases or other health conditions as medical marijuana. Although there have been many reports of marijuana’s potential therapeutic effects, the FDA has not approved marijuana as a medicine and the National Institute on Drug Abuse (NIDA) does not recommend using marijuana joints for therapeutic purposes. Some of the reasons cited are that it is an unpurified plant with a cognitive impairing property that, when smoked, is a known health hazard.
Despite this, marijuana is often used to treat a variety of problems, and many states have legalized medical marijuana use. Common uses for marijuana include treatment of intraocular pressure in glaucoma (due to vasodilation of blood vessels in the conjunctiva), suppression of nausea and vomiting during chemotherapy, stimulation of appetite in cachexia (a type of ongoing muscle loss), and relief from multiple sclerosis symptoms. One of the most commonly reported uses of medical marijuana is for pain relief, especially neuropathic pain that is not as easily treated by traditional pain relievers.
Medical Marijuana: Get the Facts [2:37]
Because cannabis that is low in THC is non-psychoactive, many medical uses of cannabis use hybrid strains that contain high amounts of CBD. Strains that are CBD-rich have equal or more amounts of CBD when compared to THC, while CBD-dominant strains or products have very little THC content. After the passage of the Hemp Farming Act in 2018, cannabis that contains less than 0.3% THC is considered hemp, not marijuana, and is federally legal. Despite this, CBD derived from marijuana is still classified as a Schedule I drug.
The Differences Between Hemp & Marijuana [4:28]
Certain conditions have been reported to be responsive to CBD treatment, including schizophrenia, inflammatory disorders, social anxiety disorder, mental depression, nausea, and vomiting induced by chemotherapy. One of the first demonstrations of therapeutic CBD use was an oil extract called Charlotte’s Web, which was named after Charlotte Figi, a five-year-old child with Dravet syndrome (a rare and severe type of epilepsy) that was treated with it. To learn more about Charlotte Figi and her story, watch the video below:
CNN documentary on Charlotte’s Web, medical marijuana treating seizure disorders [7:44]
Charlotte’s Web was a hybrid containing 17% CBD and only 0.3% THC but was never an officially approved medical treatment for seizures. In 2018, the FDA approved Epidiolex®, an extract mixture of 99% CBD and less than 0.1% THC, for treatment of Dravet syndrome and another severe form of epilepsy, Lennox-Gastaut syndrome. This was the first FDA-approved drug containing a substance derived from marijuana. Epidiolex® was classified by the DEA as a Schedule V controlled substance.
Other cannabinoid medications have been released. Dronabinol (Marinol®) was a synthetic THC approved in 1985 by the FDA as an appetite stimulant and antiemetic (anti-vomiting) drug. Another cannabinoid, nabilone (Cesamet®), was approved by the FDA in 2006 to treat neuropathic pain, nausea, and vomiting. It is also a synthetic cannabinoid that mimics THC. DEA has classified nabilone and dronabinol as Schedule II and III controlled substances, respectively. Finally, in the early 2010s, several European nations approved nabiximols (Sativex®), an oral spray with approximately equal amounts of THC and CBD, for use in treating pains related to neuropathy, cancer, and multiple sclerosis. Nabiximols have not yet been approved by the FDA for use in the U.S. but have been studied in several Phase 3 clinical trials for the treatment of cancer pain.
As mentioned earlier, the FDA approved Epidiolex® (cannabidiol) as a treatment for dravet syndrome and Lennox-Gastaut syndrome, two rare and severe epileptic conditions. This was the first time the FDA had approved a purified drug substance derived from marijuana
With the passage of the Hemp Farming Act of 2018, CBD was legalized if it was derived from industrial hemp but remained classified as a Schedule I controlled substance if it was derived from marijuana hemp.
Chapter Review and Summary
In this chapter, we learned about the cannabis plant and its two main components, THC and CBD, and compared the two. We covered the history of cannabis and marijuana use before moving on to the endocannabinoid system, where endocannabinoids use the retrograde transmission to inhibit neural activity. We then covered the pharmacological properties of THC and described its pharmacokinetics, mechanism of action, effects, tolerance, and withdrawal. Finally, we discussed medical marijuana and the few FDA-approved cannabinoid medications.
Chapter 13 Practice Questions
Answer the following questions:
- What are the three variations of Cannabis plants? Briefly describe each.
- How much THC content is required for cannabis to be classified as hemp?
- What schedule is marijuana?
- Describe retrograde transmission. How does this differ from typical neurotransmission?
- Which route of administration has a higher bioavailability: inhalation or ingestion?
- What effects do THC and CBD have on the CB1 receptor?
- Name at least four ways that THC alters or impairs normal cognition.
- The THC content in cannabis has steadily increased in recent decades. Explain why this could cause an increase in dependency and addiction.
- Is smoking cannabis strongly associated with lung cancer? Why or why not?
- Name three FDA-approved cannabinoid medicines and their clinical uses.
- Explain the significance of the Hemp Farm Act of 2018.
2nd edition
