Chapter 11: Alcohol
2nd edition as of August 2022
Chapter Overview
To close out this unit on stimulants and depressants, we will spend the entirety of this chapter looking at alcohol. Alcohol is a CNS depressant, though it has many other pharmacological actions as well. Due to the popularity and wide use of alcohol at the college level, this chapter will go into greater detail than usual to explore its unique properties and cover the breadth of alcohol research. Stay focused, and do not forget to take breaks when you need them.
Chapter Outline
- 11.1.1. History and Legislation
- 11.1.2. Prevalence of Alcohol Use
- 11.1.3. Ethanol Properties and Concentrations
11.2. Absorption and Distribution
- 11.2.1. Absorption of Ethanol
- 11.2.2. Distribution of Ethanol
- 11.2.3. Blood Alcohol Concentration
11.3. Metabolism and Excretion
- 11.3.1. Metabolism of Ethanol
- 11.3.2. Genetic Polymorphisms of ADH and ALDH
- 11.3.3. Microsomal Ethanol-Oxidizing System
11.4. Ethanol Pharmacodynamics
- 11.4.1. Receptors and CNS Effects
- 11.4.2. Ethanol Dependence
- 11.4.3. Effects on Other Systems
- 11.5.1. Alcohol-Related Cancers
- 11.5.2. Wernicke-Korsakoff Syndrome
- 11.5.3. Fetal Alcohol Syndrome
11.6. Treatment of Alcohol Use Disorder
- 11.6.1. Alcohol Abuse vs. Alcohol Dependence
- 11.6.2. DSM-V-TR Criteria for Alcohol Use Disorder
- 11.6.3. Treatment of Alcohol Use Disorder
Chapter Learning Outcomes
- Describe the history of alcohol and its prevalence.
- Clarify the importance of ethanol.
- Describe the absorption and distribution of ethanol.
- Describe the metabolism and excretion of ethanol.
- Describe the pharmacodynamics of ethanol and its effects on the various systems of the body.
- Identify and describe adverse health risks associated with ethanol.
- Discuss how alcohol use disorder is treated.
11.1. Alcohol Overview
Section Learning Objectives
- Explain the history of alcohol use and legislation.
- Explain how prevalent alcohol use and alcohol use disorder are.
- Describe how ethanol is measured and define standard drink.
Alcohol (or more specifically, ethanol) is a drug that has a long history and is extensively used throughout the world. Before exploring the pharmacology of alcohol, we will start by learning more about its use, prevalence, legislation, chemical composition, and measurement.
11.1.1. History and Legislation
It is estimated that humans have been consuming alcohol for around 9000 years. Wine and beer existed in ancient Egyptian and Sumerian civilizations, with records dating as far back as 3200 B.C. Ancient Greeks used wine in religious ceremonies around 800 B.C., and Roman culture involved heavy drinking that persisted until the fall of Rome in 476 A.D. Distilled liquor first appeared around 1250 A.D., eventually followed by brandy, gin, and rum.
Alcohol is created by the fermentation of sugars from a variety of sources, such as grapes (wine), grains (beer), honey (mead), and sugarcane (rum). Methods of production spread throughout the world and contributed to its popularity. Alcoholic beverages were seen as more nutritious than water. Its consumption was also tied to social interaction, religious ceremonies, and celebrations, which increased its cultural importance.
Opposition to alcohol use has existed for as long as the recorded history of alcohol. In the United States, Dr. Benjamin Rush was a signer of the Declaration of Independence and was recognized by the American Psychiatric Association as the “Father of American Psychiatry”. He recognized alcoholism as a disease and was an early temperance leader. the movement to prohibit alcohol started to pick up steam in the 1800s. After the Civil War, traction for the movement increased, fueled largely by the Women’s Christian Temperance Union. The National Prohibition Act, also known as the Volstead Act, was passed in 1920 and added to the Constitution as the 18th Amendment. It banned the sale and distribution but not the consumption of alcohol across the U.S.
During the Prohibition era, consumption of alcohol was driven underground to speakeasies. At the same time, widespread smuggling of alcohol, known as bootlegging, arose to meet demand sparking an increase in organized crime. Support for prohibition steadily dropped, and 13 years later, it was repealed by the 21st Amendment in 1933. Although this marked the end of national prohibition, some counties and towns remained “dry” and continued to prohibit the sale of alcohol to this day. Individual states were assigned responsibility for regulating alcohol and setting their own legal drinking ages. The National Minimum Drinking Age Act, passed in 1984, ensured that sale was prohibited to anyone under the age of 21. Any state that did not comply with this legislation lost 10% of its federal highway funding. This was reinforced by the National Highway System Designation Act of 1995, which required that states adopt a “zero-tolerance law” to prohibit under-21 driving if the operator had at least a blood alcohol level of 0.02%.
11.1.2. Prevalence of Alcohol Use
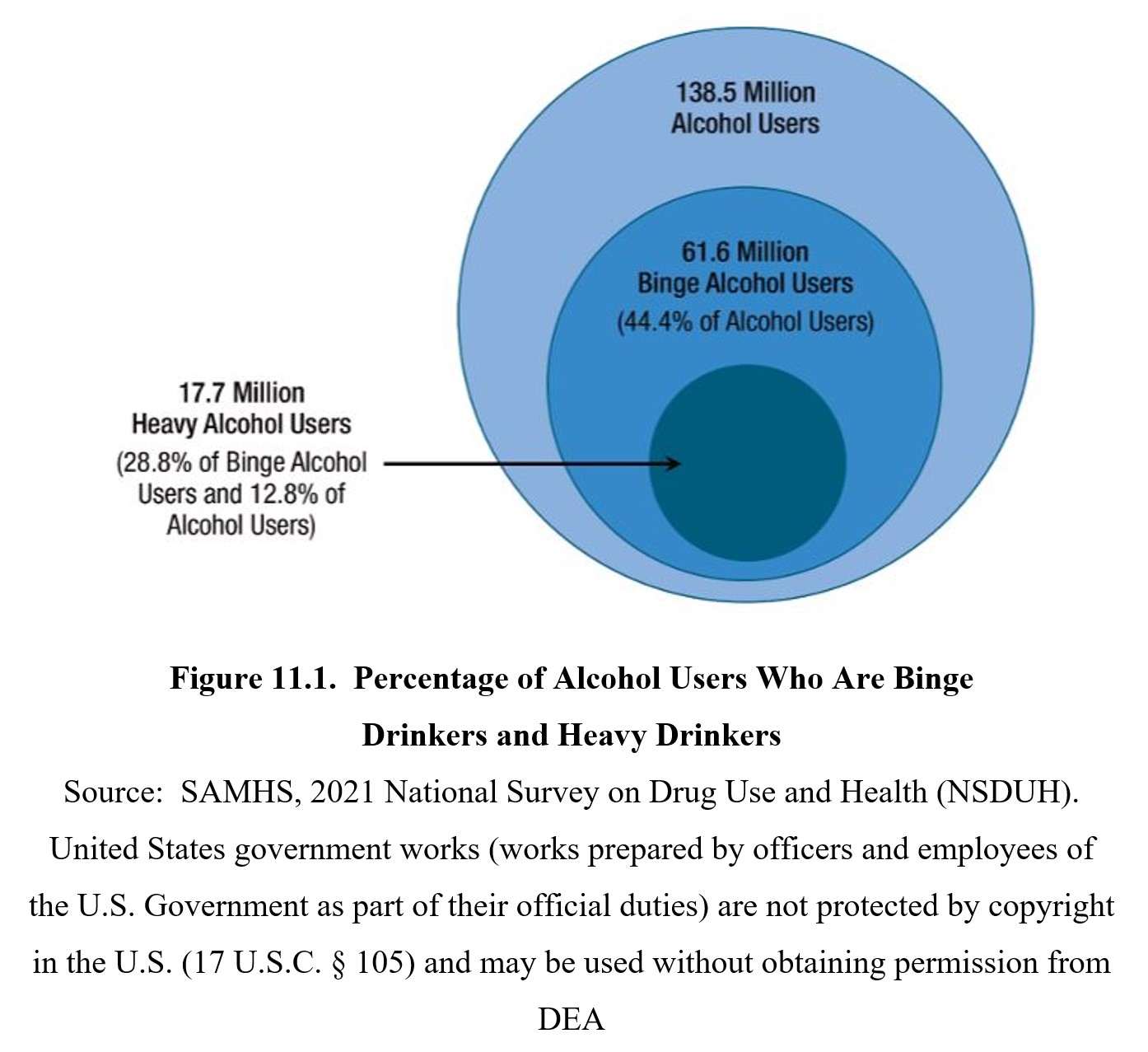
Alcohol is one of the most commonly used drugs. According to the 2020 National Survey on Drug Use and Health (NSDUH), over half of Americans aged 12 or older reported that they drank in the past month. This widespread use of alcohol also contributes to risky behaviors. Binge drinking involves drinking large amounts of alcohol in a short time span (usually 4–5 drinks in about 2 hours) and carries a higher risk than normal drinking. Roughly 23% of people 18 and older reported binge drinking in the past month (SAMHSA, 2021).
Driving while intoxicated (DWI), also known as driving under the influence (DUI), is another common consequence of alcohol use. Vehicle collisions involving alcohol-impaired driving accounted for 29% of all driving fatalities in the U.S. in 2019. Similar to smoking, alcohol consumption also imparts considerable health risks to the user. Alcohol is the third-most preventable cause of death in the U.S, after smoking and poor diet (Mokdad et al., 2004).
Underage drinking is also common. According to the 2020 NSDUH, nearly 40% of people ages 12-20 reported drinking alcohol. Although young people drink less often than adults, they also engage in riskier behaviors. More than 90% of all alcoholic drinks consumed by young people involved binge drinking. Statistics show that at least 60% of undergraduate students acknowledge past-month consumption of alcohol.
Underage Drinking (NIAAA, n.d.)
Alcohol use disorder (AUD) is a substance use disorder outlined in the DSM-5-TR. It is characterized by compulsive alcohol use despite negative consequences. The DSM-5 defines 11 criteria for diagnosis, with the severity of AUD (mild, moderate, or severe) depending on the number of criteria met. Nearly 15 million people have AUD, with AUD being more common among men (9.0 million) than women (5.5 million) (SAMHSA, 2021).
11.1.3. Ethanol Properties and Concentrations
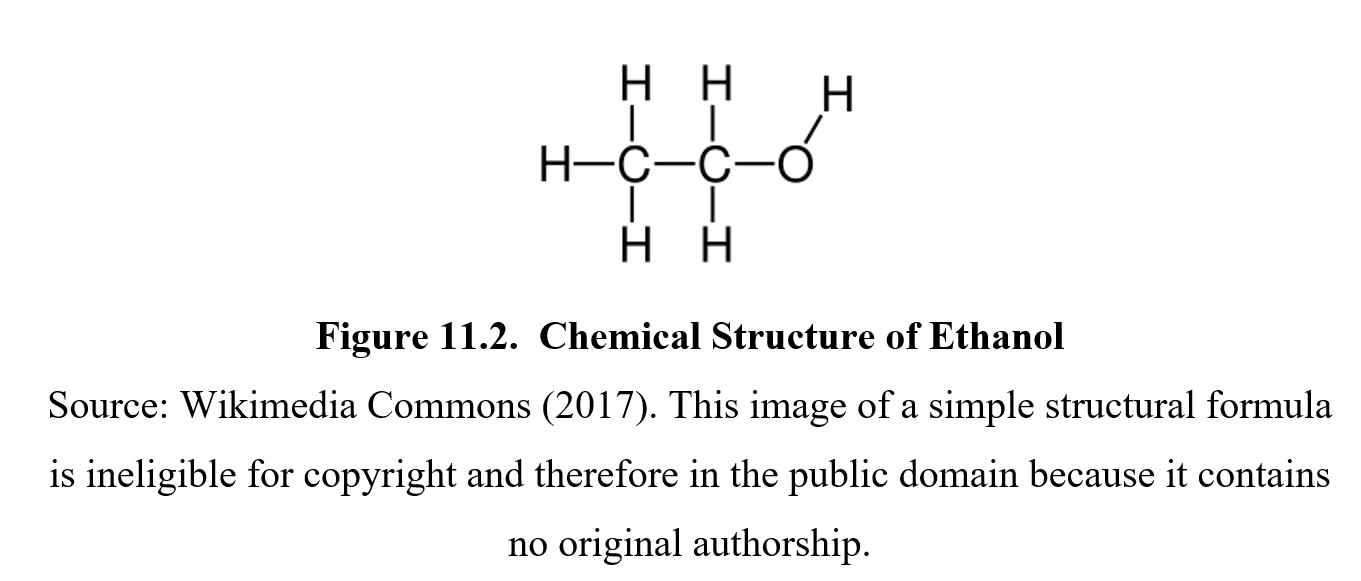
The scientific name for the drug is ethyl alcohol or ethanol. An ethanol molecule is comprised of an ethyl group (CH3−CH2−) linked to a hydroxyl (OH) group, as you can see in the images below. Because of this, the molecule is often abbreviated as EtOH.
Alcohol or Ethanol?
Although the terms alcohol and ethanol are mostly interchangeable in this chapter, we will prefer the term ethanol when talking about the drug. In chemistry, alcohol can refer to various compounds that have similar molecular structures to ethanol. You won’t usually see this meaning of the word used in the context of drugs—ethanol is the only alcohol widely consumed by humans, so the term alcohol is regularly used in the scientific literature—but it is worth being aware of the distinction. Other alcohols include isopropyl alcohol (rubbing alcohol) and methanol (wood alcohol), but these cannot be safely consumed.
Ethanol is an organic compound with a variety of applications. It is used as a disinfectant, such as in hand sanitizers or medical wipes. It is also a universal solvent and has many uses in the manufacture of other compounds. Ethanol is a suitable fuel source and is used in alternative fuels. It is also, of course, the key ingredient in alcoholic drinks.
There are multiple ways to measure the amount of ethanol in an alcoholic drink. The simplest approach is to describe the percentage of ethanol content. This is used by the standard measurement of alcohol by volume, abbreviated as ABV. Instead of ABV, you might also see alcohol proof used instead. The proof of a drink is defined as twice the percentage of ABV, and so, ranges from 0–200.
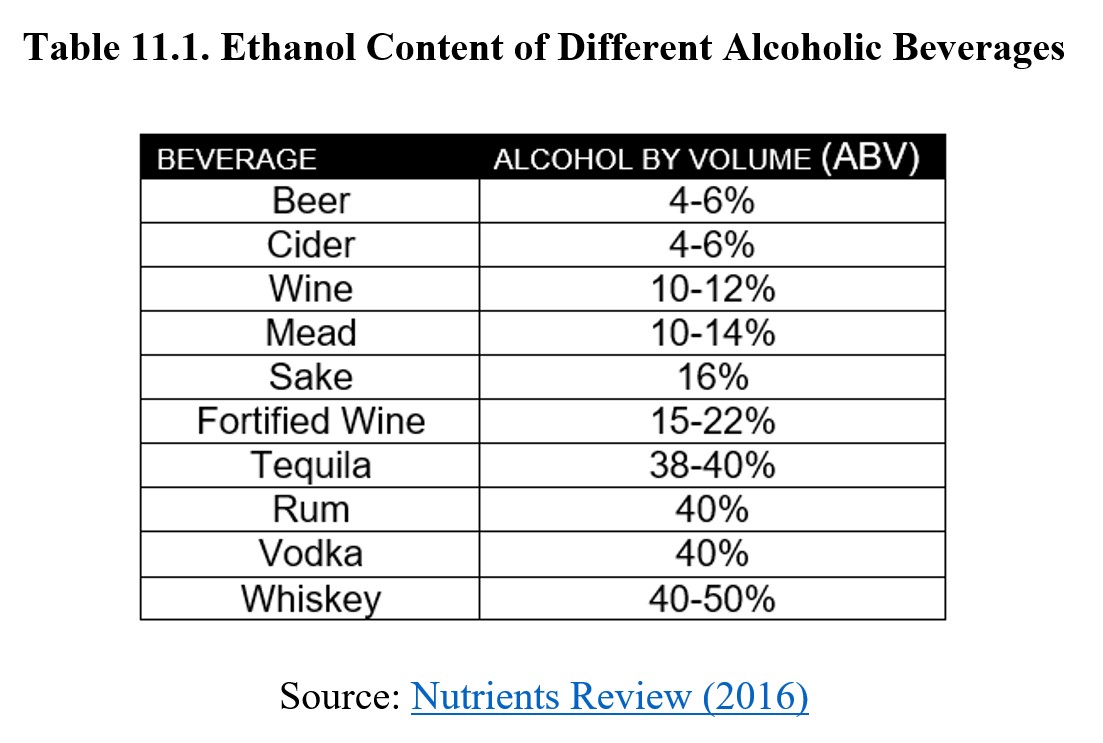
An absolute or anhydrous solution of ethanol is 100% ethanol. Another common specification is outlined by the U.S. Pharmacopeia (USP) as 95% ethanol solution. Both of these concentrations are intended more for use in research, medicine, and industry. Alcoholic drinks tend to have concentrations in the 1–20% for beers and wines, with distilled spirits usually having around 40% ABV. Consult the table below to view typical percent ABV for various alcoholic beverages:
A common shorthand for determining alcohol consumption is a standard drink or an alcoholic drink equivalent. A standard drink contains about 0.6 fluid ouches or about 14 grams of pure ethanol. This amount of alcohol would be contained within a 12-oz can of beer (around 5% ethanol), a 5-oz glass of wine (around 12% ethanol), or a 1.5-oz shot of distilled spirits (upwards of 40% ethanol). Three standard drinks of any of these alcoholic beverages will usually put you over the legal limit for driving, though there is individual variability and even a single drink in some individuals can cause significant impairment.

11.2. Absorption and Distribution
Section Learning Objectives
- Describe how ethanol is absorbed and what factors influence the rate of absorption.
- Explain how ethanol is distributed throughout the body and how individual characteristics can influence blood concentrations.
- Define blood alcohol concentration and describe the effects of alcohol at different BACs.
The pharmacokinetics of ethanol starts with its absorption and distribution. These topics are important to more than just neuroscience and health professional students. Understanding how alcohol is absorbed is necessary for responsible drinking. Laws that are written to punish drunk driving reference the concentration of ethanol in the blood.
11.2.1. Absorption of Ethanol
Ethanol is extremely soluble in both water and fat, making it readily absorbed through tissue. Ethanol is typically ingested orally through alcoholic drinks. These can also be administered rectally, although the practice is uncommon and not advised. Alcohol enemas results in faster absorption and higher blood concentrations because it bypasses the stomach. Another practice called vodka eyeballing involves introducing vodka into the eye sockets. Contrary to popular belief, eyeballing vodka does not induce rapid intoxication and runs the risk of corneal abrasions and scarring.
When taken orally, only 20-25% is absorbed by the stomach, while 75–80% is absorbed through the small intestine. The oral bioavailability of ethanol is poor for two reasons. First, there are alcohol-metabolizing enzymes found in the stomach wall. Second, any ethanol that is absorbed from the digestive tract enters the hepatic portal vein which goes straight to the liver. This will be explained further in the section on Metabolism of Ethanol.
The presence of food in the stomach delays transit from the stomach to the small intestine. This results in stomach enzymes having a longer time period to oxidize ethanol before it can enter the small intestine. Consequently, food reduces peak blood concentration. This is the reason why eating food while drinking is advised. Refer to the graph below to see the effect of food on ethanol absorption:
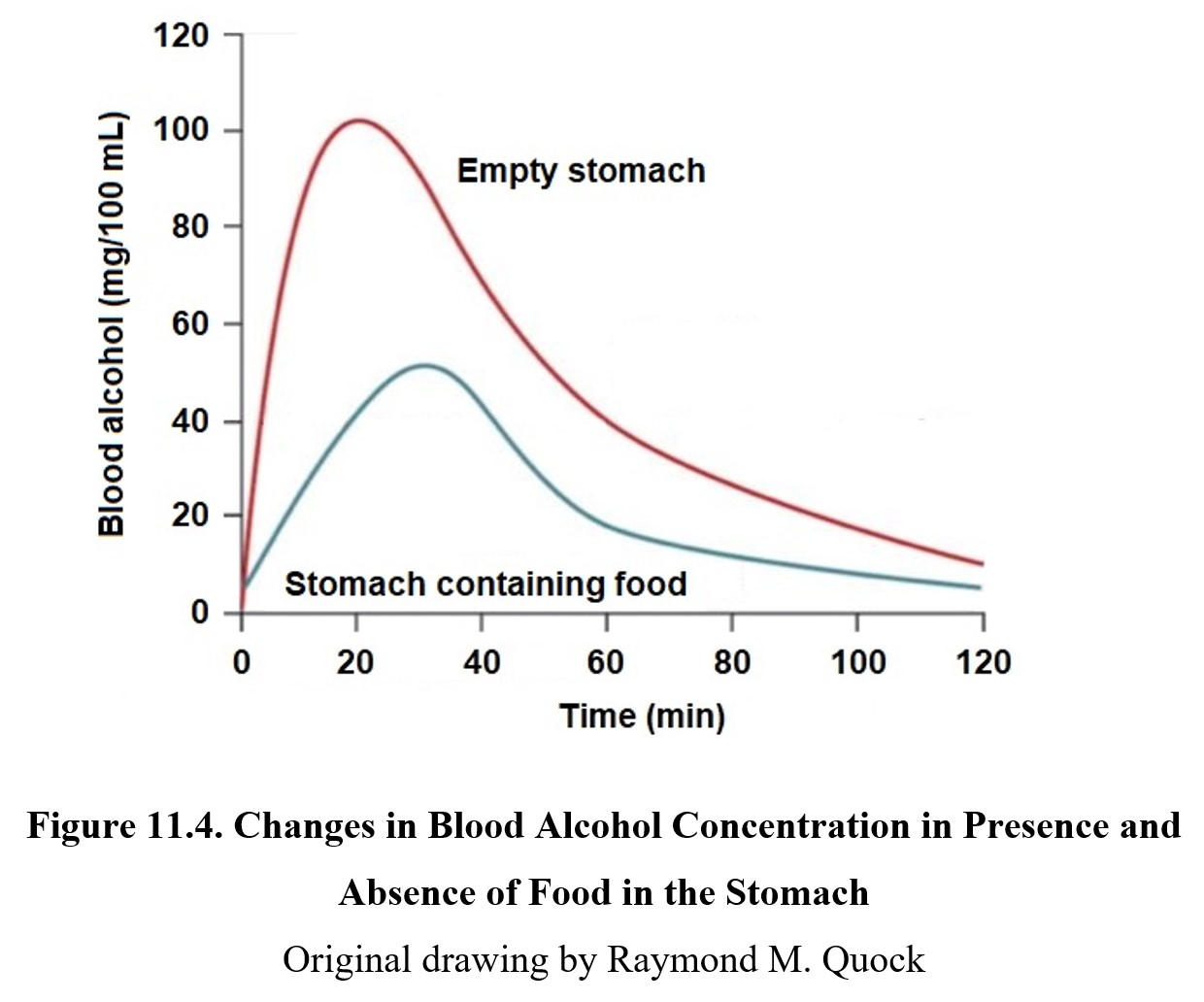
On an empty stomach, 50% of ethanol is absorbed in 15 minutes, with maximum blood levels being reached in 20 minutes. Most of the ethanol (80–90%) is absorbed within an hour or less of administration. The more food in the stomach, the less ethanol absorbed, although there is a gender difference. There is a gender difference in the activity of stomach and liver enzymes so that a larger amount of ethanol will be absorbed by women than men.
11.2.2. Distribution of Ethanol
Ethanol is miscible with water, meaning that when the two are mixed, ethanol is distributed evenly throughout the solution. This means that once ethanol is absorbed, it is found in approximately equal concentrations in water throughout the body. This includes not only blood but also the water in tissues and other fluids. Altogether, this is known as the total body water.
The amount of total body water varies depending on age and gender, which influences the concentration of ethanol. Women tend to have a higher percentage of body fat and thus a lower percentage of body water than men. Total body water also tends to decrease with age. The lower the total body water, the more concentrated is the ethanol, so the same amount of alcohol will produce greater effects in women and older people. Compare the percentage of total body water in different types of people in the table below:

Because it is both water- and fat-soluble, ethanol can also cross barriers very easily. Ethanol crosses the blood-brain barrier to reach synapses in the brain. It is also able to cross the placenta in pregnant women to reach the fetus, which can result in developmental disorders. We will discuss this in more detail later in this chapter in the section on adverse health risks.
11.2.3. Blood Alcohol Concentration
The concentration of ethanol in the body is measured as blood alcohol concentration (BAC), also called blood alcohol content or blood alcohol level (BAL). BAC is defined as the number of grams of ethanol per 100 mL of blood. For example, a BAC of 0.05 is equivalent to 0.05 grams of ethanol per 100 mL of blood.
The legal limit for BAC while driving is 0.08, except in Utah where it is 0.05. For drivers under 21 years of age, most states have penalties for BAC above 0.02, although some states have stricter laws. BAC can be estimated by breathalyzer tests, which measure the amount of ethanol expelled through respiration. Although this is not identical to actual blood alcohol concentration, it is reasonably close in most circumstances and can be used as a basis for arrest.
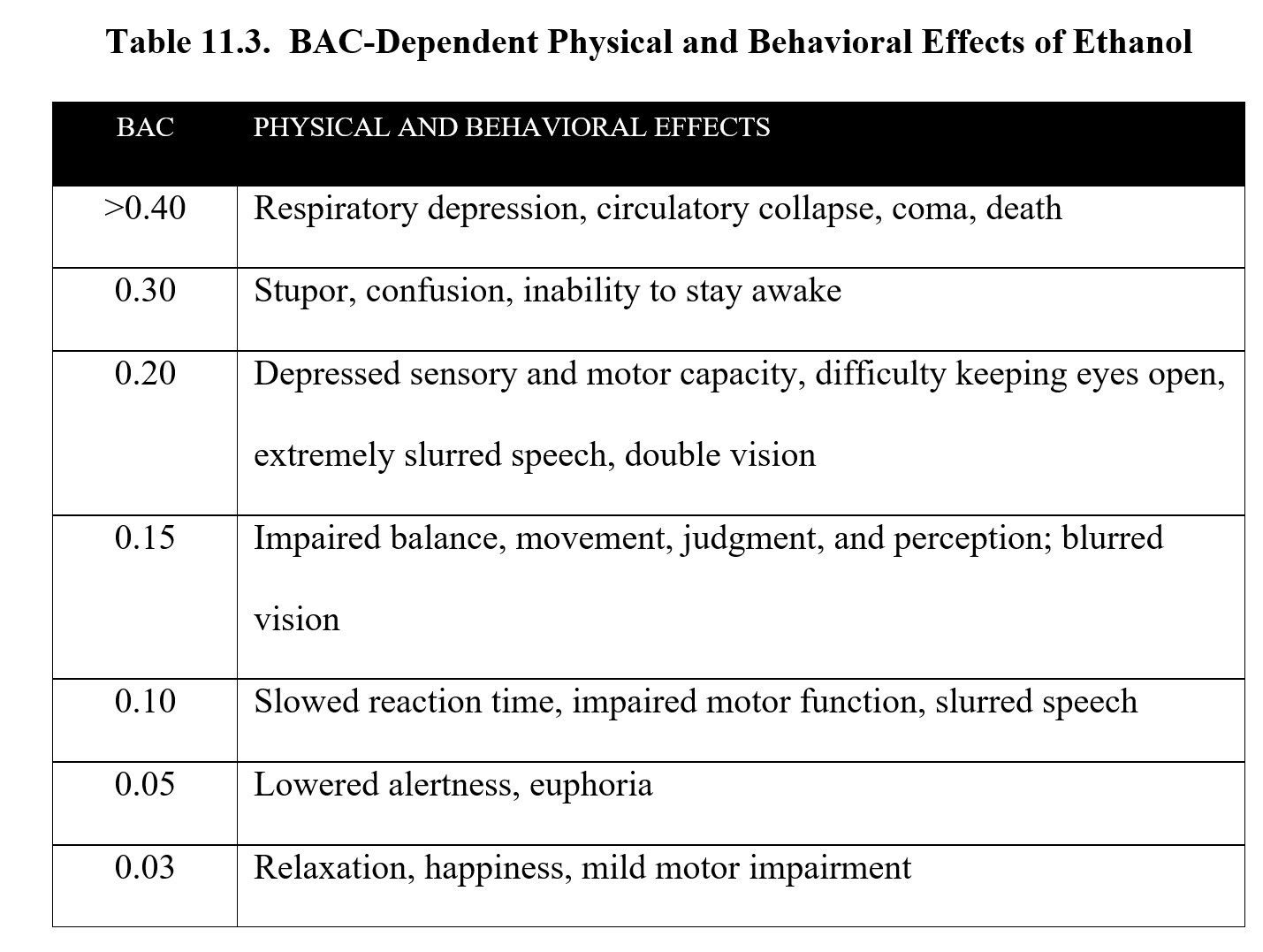
Below is a table of the effects of alcohol at different BACs. As you can see, although the legal limit is 0.08, impairment occurs at lower concentrations. Take a moment to study the contents of the table before moving on.
Blood Alcohol Concentration [5:27]
11.3. Metabolism and Excretion
Section Learning Objectives
- Explain the kinetics of ethanol elimination.
- Describe how ethanol is metabolized and discuss differences in metabolism by gender.
- Explain genetic polymorphisms and describe the effects and prevalence of atypical ADH and ALDH enzymes.
- Explain how disulfiram is used in the treatment of alcohol use disorder.
- Describe the microsomal ethanol-oxidizing system and its effect on ethanol kinetics.
A portion of ethanol is metabolized in the stomach before it is absorbed into the hepatic portal vein. The hepatic portal vein then delivers the absorbed ethanol to the liver before it is distributed into the systemic circulation. Ethanol metabolism is a complex topic, so we will devote more time than usual to address it. This was described in the section on first-pass metabolism in the chapter on Pharmacokinetics. There will be a lot of new terms in this section, so make sure to pause and check your understanding every so often.
11.3.1. Metabolism of Ethanol
Most of the ethanol ingested (around 90–95%) is metabolized by the body. The remainder is excreted as is in urine, feces, and other secretions. Some of the non-metabolized ethanol escapes via the lungs, which is why breathalyzers can measure BAC indirectly.
Ethanol is eliminated by zero-order kinetics. All the drugs we have covered so far followed first-order kinetics and have half-lives. Ethanol does not have a half-life because a fixed amount of it is metabolized per unit of time (note the relatively flat and parallel slopes of the graphs below). Therefore, drinking more alcohol does not speed up metabolism—with a few notable exceptions that we will get to later in this section. Typically, it takes about an hour for the body to clear itself of one standard drink of alcohol (i.e., 12-oz beer, 5-oz wine, or 1.5-oz distilled spirits).
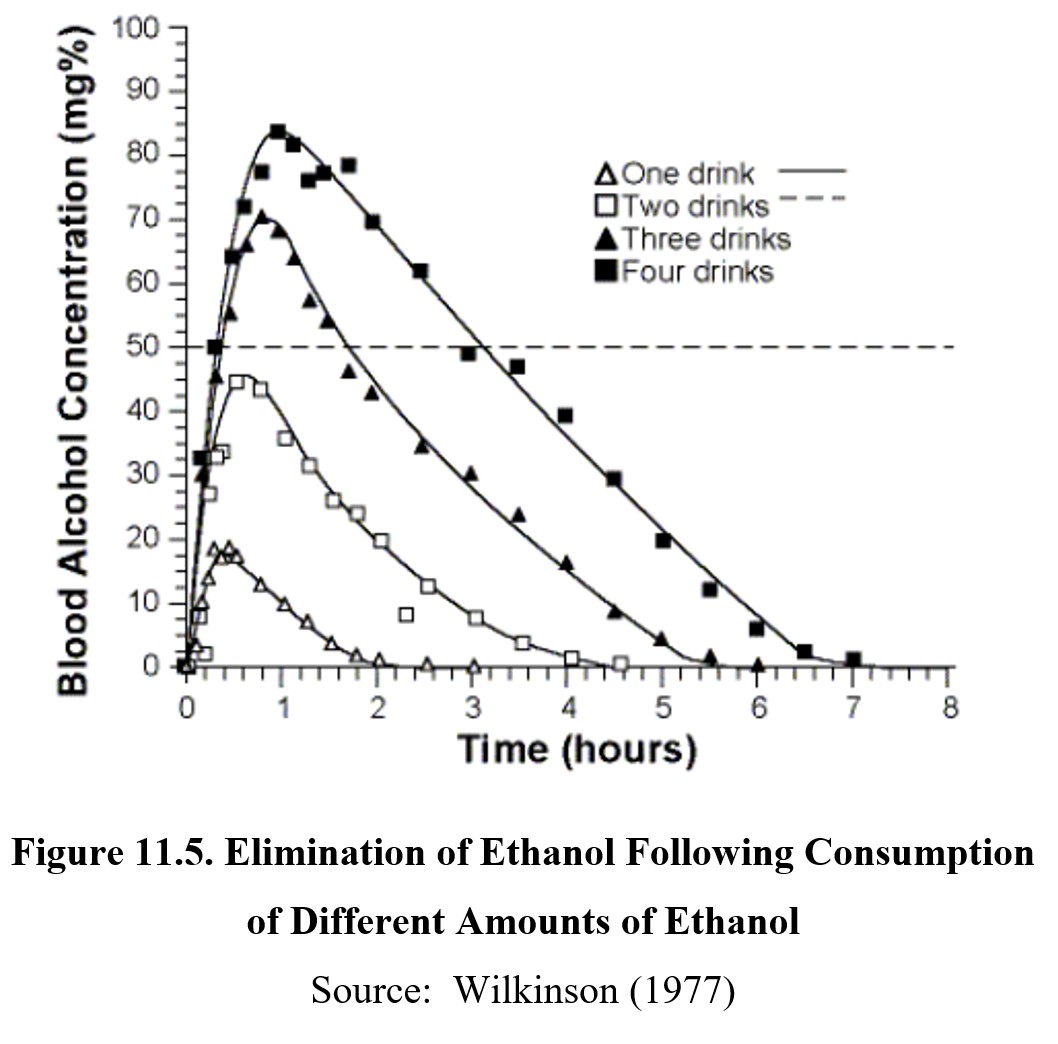
Metabolism of ethanol follows two important steps. The first step involves oxidizing ethanol in acetaldehyde. Acetaldehyde is a toxic substance that contributes to the negative symptoms associated with hangovers and is a known carcinogen. The enzyme that catalyzes this reaction is alcohol dehydrogenase (ADH) and is found in the stomach and liver. The activity of ADH is rate-limited, making ADH the reason why ethanol follows zero-order kinetics.
Acetaldehyde is then rapidly converted to acetic acid by aldehyde dehydrogenase (ALDH). The metabolite produced, acetic acid, is much less harmful than acetaldehyde and is eventually broken down into carbon dioxide and water.

Organizing Key Terms in Ethanol Metabolism
Before moving on, let’s address a practical matter—distinguishing between these terms can be difficult without a background in chemistry. Still, this is key information that needs to be memorized. To distinguish the terms and remember their meanings, it may be useful to use logic to organize them and place them in order.
First, remember that alcohol is metabolized by alcohol dehydrogenase. This should be simple because they both have referred to alcohol by name. Next, note how acetaldehyde is metabolized by aldehyde dehydrogenase. Acetaldehyde is an aldehyde, which is why it is included in both terms.
In a similar vein, acetic acid is a metabolite of acetaldehyde. Finally, remember that ADH comes before ALDH in the process because it has fewer letters. These patterns and mnemonics should help you link these terms together.
Ethanol metabolism differs by gender. Overall, men metabolize more ethanol than women. In men, ADH is present in the stomach lining and can reduce ethanol absorption by up to 30%. In comparison, women have little to no ADH in their stomach walls. Women have less ADH in the liver as well. Between these differences and the differences in total body water mentioned in the last section, women get higher BACs than men after ingesting the same amount of ethanol.
11.3.2. Genetic Polymorphisms of ADH and ALDH
In biology, you probably learned about DNA, genes, and mutations. Mutations occur when a section of DNA gets randomly changed. The change may be beneficial, harmful, or even fatal, but mutations are rare occasions. A genetic polymorphism is like a mutation, except the changes appear more often in the population (at least 1%). One type of polymorphism, called a single nucleotide polymorphism (SNP), occurs when a single unit of DNA, known as a nucleotide, is substituted with a different nucleotide. For example, guanosine is the most frequent nucleobase at a specific base position in the genome, but there is a SNP in which the guanosine is replaced by adenine.
Genes, or functional sections of your DNA, are responsible for enzyme expression. In this context, you can think of a gene as a set of instructions for how to manufacture a particular enzyme. When a gene has a polymorphism (SNP), those instructions may be altered. This can result in an enzyme with a slightly different characteristic.
There are two enzymes involved in the metabolism of ethanol in which polymorphisms have been discovered and intensely studied by scientists, ADH and ALDH. These polymorphisms will determine drinking habits and individual susceptibilities to alcohol toxicity and alcohol abuse.
The ADH enzyme is actually encoded by seven genes on Chromosome 4 but exists in five different classes of ADH enzyme in humans, labeled ADH1, ADH2, etc. An allele is a variant of the DNA sequence found at the same locus on a chromosome. The ADH enzyme is polymorphic, and the gene that encodes ADH2 has SNPs that produce several variants of the ADH2 enzyme. The typical enzyme is listed as ADH2*1, while the variants are ADH2*2 and ADH2*3. Examine the table below to see how they compare:

As you can see, the variants metabolize ethanol at a faster rate than normal. Although this sounds like an advantage, remember that ADH converts ethanol to acetaldehyde. If the ADH catalyzes at a faster than normal rate, this results in a buildup of toxic acetaldehyde. This can cause flushed skin, headaches, and a sensation of warmth. Because there is less of the desired ethanol, drinking is less pleasurable. Because more of the toxic acetaldehyde is produced, people with fast-acting ADH2 tend to drink less and have a reduced risk of AUD.
Similar to ADH, there are multiple ALDH enzymes in humans—a whopping 19 have been discovered, some with names like ALDH1L2 and ALDH18A1. Fortunately for us, we only care about the ALDH2 enzyme. The gene encoding this enzyme is polymorphic, with one major variants of relevance to alcohol toxicity (see table below):

The atypical ALDH2*2 enzyme metabolizes acetaldehyde very slowly. In fact, it is so slow that it is nearly inactive. Similar to the atypical ADH enzymes, this causes a buildup of acetaldehyde and the associated symptoms, except significantly worse. As a result, people with ALDH2*2 tend to have very low rates of alcohol use and AUD.
Comparing the two tables above, you may notice that Asians are at an increased risk of both ADH2*2 and ALDH2*2. They are prone to exhibiting the “Asian flush” when consuming alcohol. Because of this, Asians as a whole tend to exhibit lower rates of alcohol use and AUD, and those who have both, experience extreme acetaldehyde toxicity. Of course, Asians are a broad group, so there are some differences by region and culture as well.
One final note about ALDH: some drugs, such as disulfiram (Antabuse®), are capable of blocking ALDH enzymes. When these drugs are administered, they cause the same effects as ALDH2*2, causing a surge in acetaldehyde levels and unpleasant hangover symptoms. As such, these drugs can be used in the treatment of AUD to punish relapse and make it less likely to occur again in the future.
11.3.3. Microsomal Ethanol-Oxidizing System
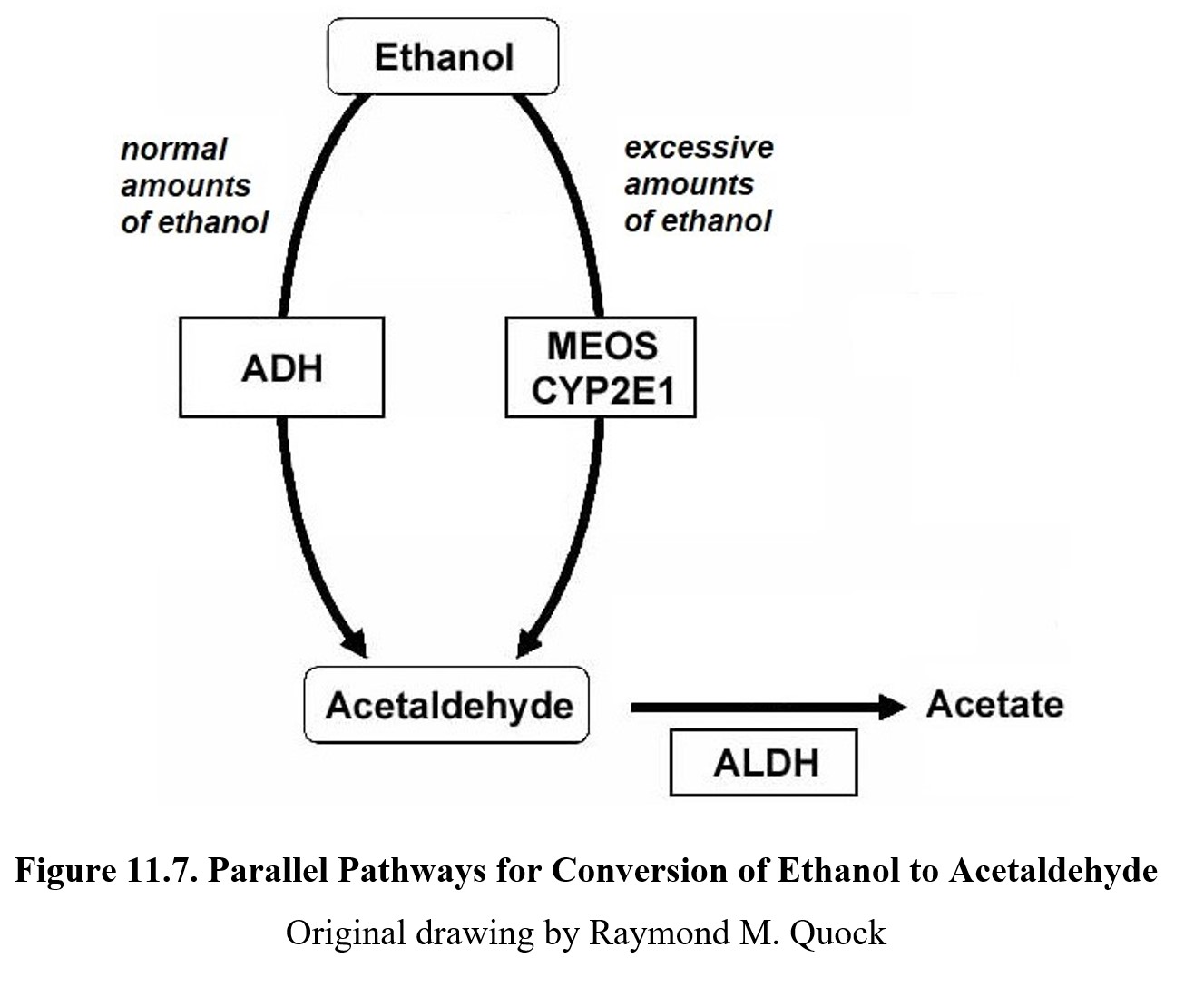
There is one final complication to ethanol metabolism that we need to cover. (I know—as if it wasn’t complicated enough already, right?) So far, when we have talked about ethanol metabolism, we have assumed that all alcohol is metabolized by ADH. This is not always true. When ADH is saturated, the microsomal ethanol-oxidizing system or MEOS is activated.
The MEOS uses part of the cytochrome P450 enzyme family, in particular, CYP2E1, to oxidize excess ethanol into acetaldehyde, the same as ADH (see image below). Unlike ADH, however, the MEOS follows first-order kinetics. This means that as BAC increases, more and more ethanol is oxidized by the MEOS, and overall metabolism is increasingly first-order. ADH is still functioning at maximum capacity during this time, so the rate is a mix of zero- and first-order kinetics.
11.4. Ethanol Pharmacodynamics
Section Learning Objectives
- Describe the pharmacodynamics of ethanol and its effect on the central nervous system.
- Explain ethanol tolerance, dependence, and withdrawal.
- Describe the effects of ethanol on other systems in the body.
Now that we have covered the pharmacokinetics of ethanol, it is time to move on to its pharmacodynamics. Ethanol has a wide range of effects beyond just CNS depression. We will begin this section by examining its mechanisms of action before moving on to dependence and effects on other systems.
11.4.1. Receptors and CNS Effects
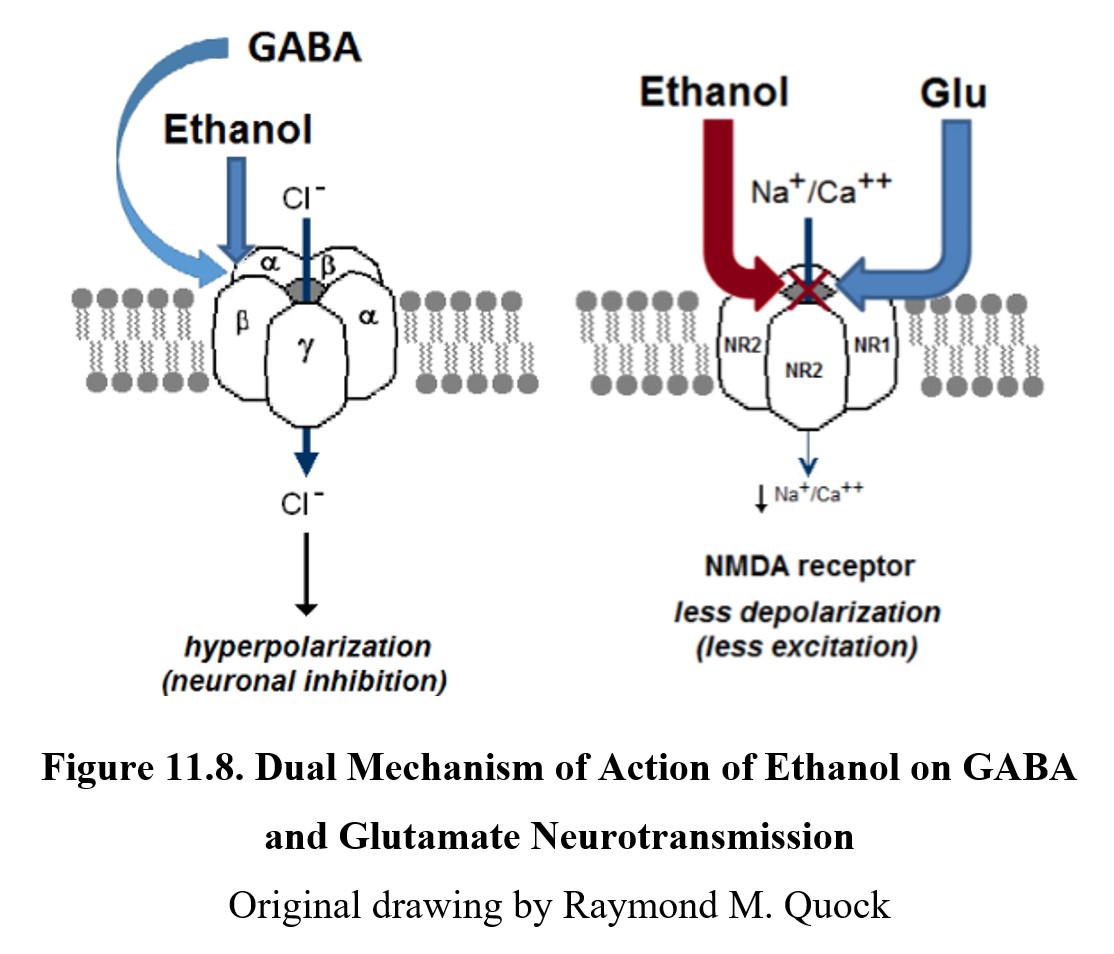
Ethanol is considered a “dirty drug,” an informal term meaning that it affects many different receptors in the body and can produce a wide range of effects. The primary depressant mechanism of ethanol occurs at the GABAA chloride channel receptor complex. The exact site that ethanol binds to is unknown, as is its exact effect on chloride ion channels, but it enhances the effects of GABA and causes hyperpolarization.
CNS depression may also be the result of ethanol binding to and inhibiting NMDA receptors for glutamate. This reduces the inflow of sodium and calcium ions into the cell. This lowers the excitatory effect of glutamate on NMDA receptors and enhances neuronal inhibition in the CNS. See the images below to compare these two mechanisms:
The effects of ethanol are biphasic. At low doses, consumption of alcohol can elicit relaxation, giddiness, and euphoria. This is known as a buzz and is what makes initial drinking feel good. As BAC rises, depressant effects start to kick in. Motor coordination is impaired, speech becomes slurred, and sensory input is diminished. At high doses, ethanol can induce stupor, sleep, respiratory depression, coma, and death. (For a full list of effects at different BACs, check the table under the BAC subsection 11.2.3 above.)
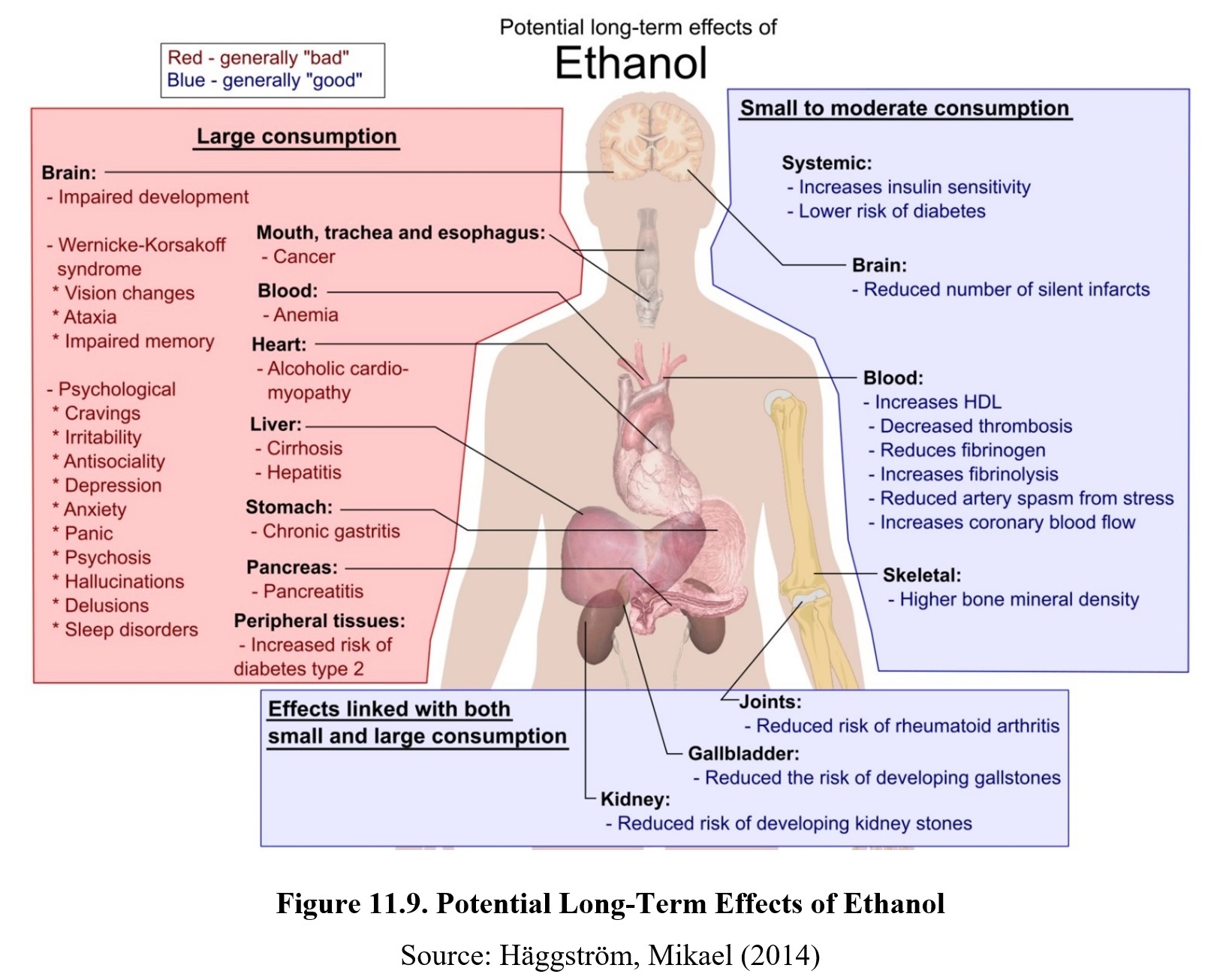
Ethanol interacts with many drugs due to its widespread influences on different receptors and their pharmacological properties. You may have noticed that many of the drugs we have covered so far have adverse interactions with ethanol. Indeed, it is the most important drug interaction in the CNS and often causes side effects when paired with medications and recreational drugs. Some of the common effects of drug interactions with ethanol are dizziness, nausea and vomiting, coma, and death.
There is another important note about ethanol drug interactions. Ethanol is a depressant drug, and its ability to cause CNS depression can be reversed by a stimulant drug such as caffeine. However, ethanol produces multiple effects that have different susceptibilities to reversal. A prominent drug effect that contributes to drunk driving is psychomotor impairment, in which mental efforts and reflexes are slowed. Sedation is reversed by hot, black coffee, but psychomotor impairment is not.
11.4.2. Ethanol Dependence
Chronic ethanol use results in both tolerance and dependence. Tolerance develops in multiple ways. Standard pharmacodynamic tolerance occurs, which involves changes in target receptor regulation (neuroadaptation). Long-term exposure to ethanol causes the receptors to downregulate and become less responsive.
Pharmacokinetic tolerance also develops with heavy drinking. Recall that when ADH is saturated, the MEOS kicks in and CYP2E1 starts oxidizing excess ethanol. Unlike ADH, CYP2E1 and other cytochrome P450 enzymes can be induced, which results in faster metabolism of ethanol. This only occurs when ADH is saturated, so this type of tolerance only affects binge drinkers and other heavy users.
Finally, becoming familiar with the effects of inebriation leads to behavioral tolerance. This is a conditioned response to the drug that allows the user to compensate for its behavioral effects. People with behavioral tolerance can appear sober even after drinking a significant amount of alcohol and may have better control over their speech, movement, appearance, and emotional state compared to inexperienced drinkers.
Like other drugs, ethanol causes reward and reinforcement by altering dopamine transmission in the reward system. Normally, dopaminergic neurons in the VTA are under tonic inhibition by GABA interneurons. This means that ambient GABA activity somewhat inhibits these neurons from firing and releasing dopamine in the nucleus accumbens.
When ethanol is ingested, β-endorphin is released from an opioid nerve terminal. β-Endorphin, in turn, activates μ-opioid (mu-opioid) receptors on the GABA interneurons and inhibits the tonic release of GABA. This, in turn, depolarizes the dopaminergic neurons and increases dopamine release in the nucleus accumbens. This is the mechanisms that underlies the reward and reinforcing effects of ethanol. See the image below for the full process:
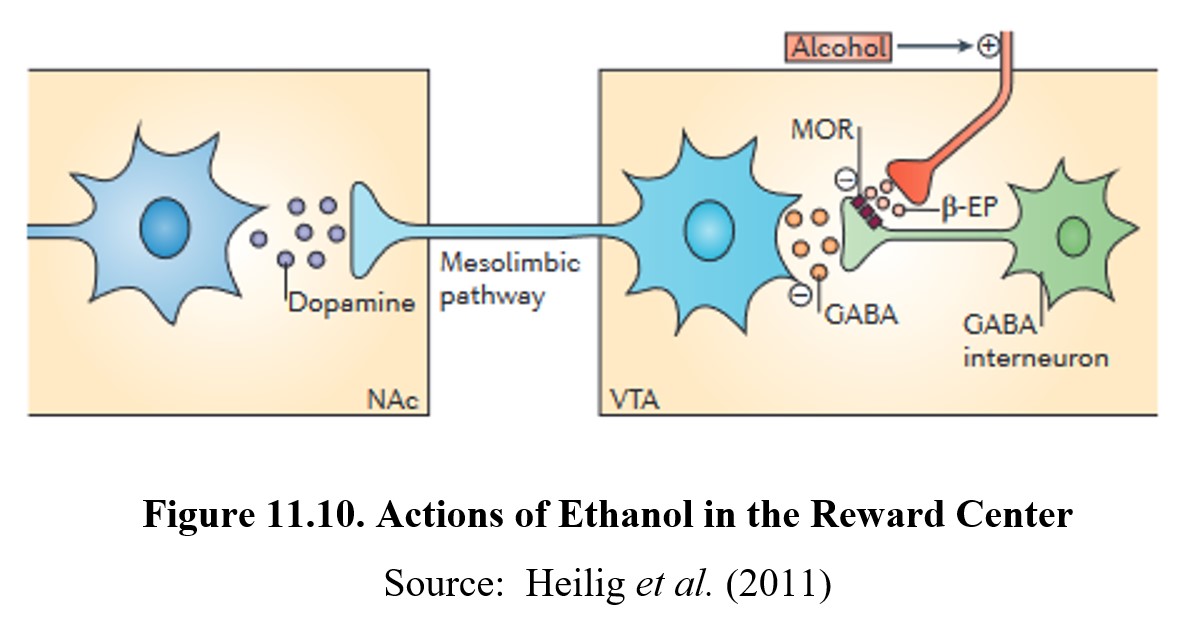
Ethanol withdrawal can be very dangerous and can last for days. Because chronic use decreases the number of GABAA receptors and increases NMDA receptors, CNS activity is significantly increased during withdrawal. Symptoms can include seizures, agitation, anxiety, increased heart rate and blood pressure, insomnia, nausea and vomiting, and paranoia. Again, we observe that signs of withdrawal proceed in the opposite direction from the effects of the drug. The intensity of the withdrawal is dependent on the duration and degree of physical dependence. During severe withdrawal, delirium tremens may occur, which involves shaking, confusion, and hallucinations and may lead to death. Delirium tremens usually occurs a few days after the onset of withdrawal and may last for two to three days.
Because ethanol withdrawal is so dangerous, treatment is usually intensive and requires hospitalization. Sedatives such as benzodiazepines are administered to mitigate withdrawal symptoms. Of those with severe symptoms, up to 15% may die from withdrawal.
11.4.3. Effects on Other Systems
Ethanol has considerable effects on systems in the body outside of the CNS. The first and foremost is the liver. The majority of ethanol is metabolized in the liver, which leads to a buildup of acetaldehyde in the liver. As mentioned before, acetaldehyde is toxic and can cause damage to the liver. Consuming large amounts of alcohol regularly will lead to a gradual deterioration of liver functioning known as alcoholic liver disease.
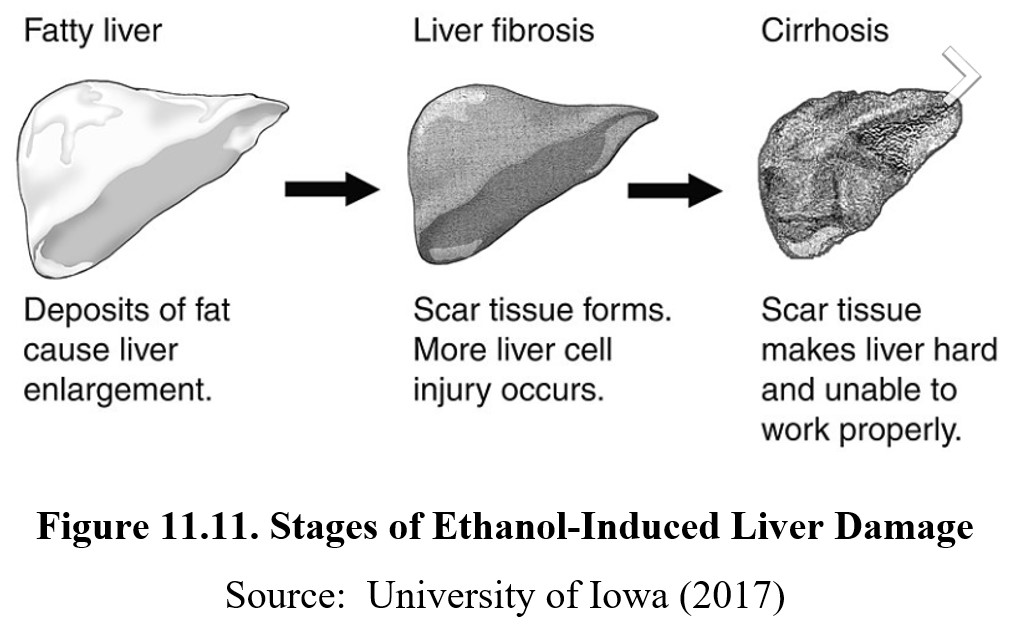
The initial stages of liver disease involve a buildup of excess fat in the liver, known as fatty liver disease or hepatic steatosis. Fatty liver disease can be reversed with changes to alcohol consumption and diet, but if consumption continues, scar tissues may form in a process known as fibrosis. The scar tissue interferes with normal liver functioning and is permanent. When large amounts of scar tissue develop, the person now has cirrhosis. At this stage, the scar tissue has grown and is killing healthy liver cells. The process is irreversible, and prolongation of life may require a liver transplant.
Ingestion of ethanol also affects the gastrointestinal system in a variety of ways. Low doses of ethanol accelerate gastric emptying, while moderate consumption stimulates digestive juices and acid secretion. At high doses, ethanol damages the lining of the stomach and causes inflammation, while also reducing the digestive system’s ability to absorb nutrients. This is the reason why physicians advise ulcer patients not to drink. Ethanol can impair the healing of the ulcer and may cause the ulcer to get worse.
The endocrine system, which is a series of glands that secrete hormones, is also altered by ethanol use. Ethanol increases blood sugar levels, which eventually results in the over-production of insulin and a subsequent decrease in blood sugar. If the patient is diabetic and receiving therapy with oral hypoglycemic drugs, ethanol is likely to complicate efforts to maintain normal blood glucose levels.
Ethanol interferes with the synthesis of Vitamin D, a hormone essential for absorption of calcium from the digestive tract. Without Vitamin D, the body’s calcium pool is diminished, affecting bone growth and remodeling. This can result in decreased bone density (osteoporosis) and an increased likelihood of broken bones.
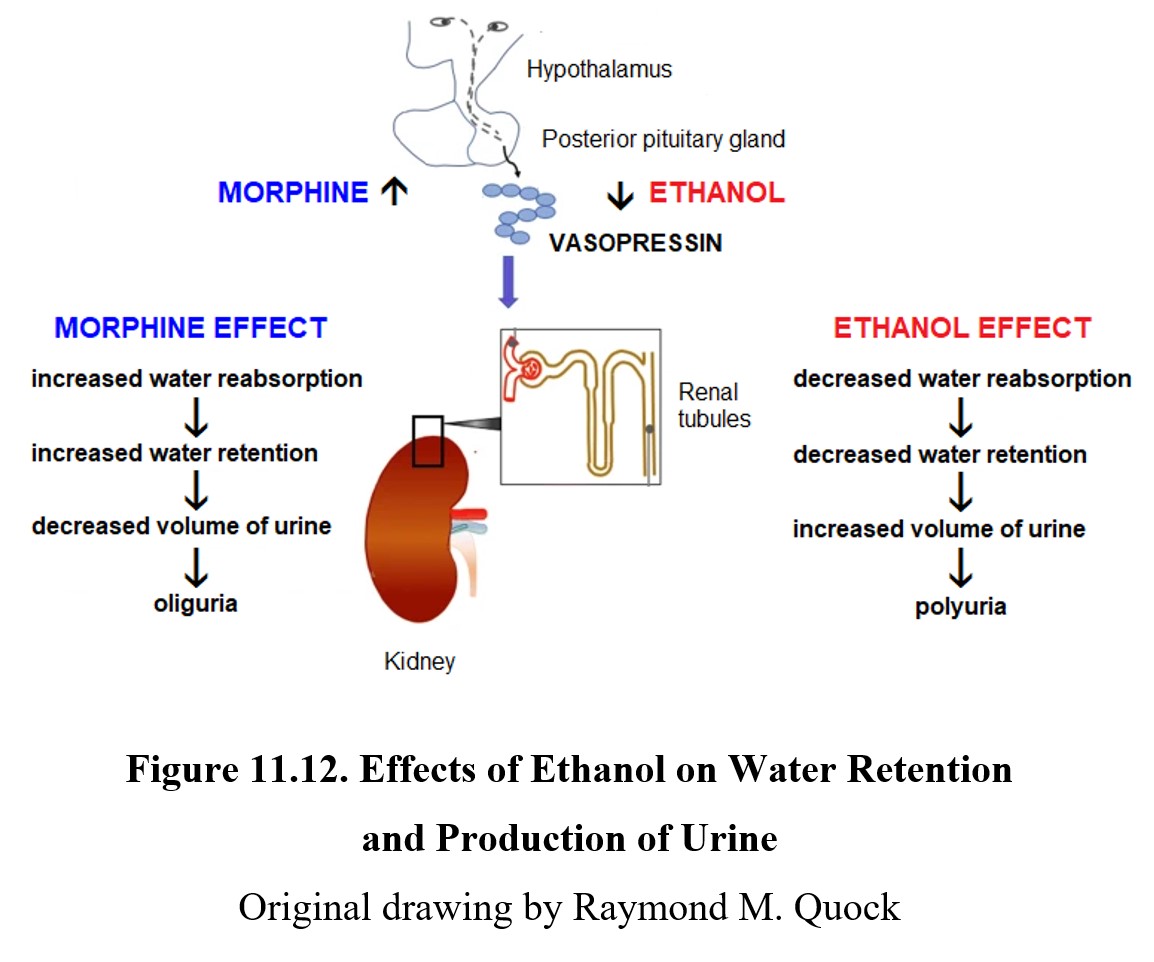
Ethanol inhibits the release of vasopressin from the posterior pituitary gland. Vasopressin normally promotes water retention by the kidney and regulates the production of urine. In the absence of vasopressin, water is not reabsorbed back into the body and is eliminated as urine. This results in polyuria or the production of abnormally large volumes of dilute urine.
Ethanol suppresses testosterone production in males by interfering with gonadotropin release from the pituitary gland. Reduced testosterone also reduces the sperm count and causes erectile dysfunction.
The effect of ethanol on women’s health is also complicated. Ethanol can increase estrogen levels but can increase the risk of estrogen-dependent breast cancer in pre-menopause women. Ethanol also reduces progesterone levels which disrupts the normal menstrual cycle and causes low libido and hot flashes. In post-menopausal women, ethanol may reduce risks for coronary heart disease and osteoporosis.
Finally, excessive ethanol consumption may suppress the immune system. This manifests as a decrease in T cells, increased memory T cells, and higher rates of induced cell death. A weakened immune system causes increased susceptibility to bacterial infections such as pneumonia, tuberculosis, hepatitis, and septicemia.
11.5. Adverse Health Risks
Section Learning Objectives
- Explain what cancers are associated with alcohol use.
- Describe Wernicke encephalopathy and Korsakoff syndrome.
- Describe the causes and symptoms of fetal alcohol syndrome.
To wrap up this chapter, we will consider some of the adverse health risks associated with ethanol use. Of course, some of the effects mentioned in the previous section are related to the risks mentioned here. While this is not a comprehensive list, it should give you some idea of how alcoholism can have far-reaching consequences beyond addiction.
11.5.1. Alcohol-Related Cancers
According to the U.S. Environmental Protection Agency (EPA), acetaldehyde is considered a possible carcinogen. Countless studies have linked the consumption of alcohol to increased risks for developing various cancers. This link is mediated by the level of exposure to acetaldehyde caused by the metabolism of ethanol.
The risk of cancer increases in all areas of the gastrointestinal tract, such as the mouth and upper throat, esophagus, stomach, and bowels. The larynx is also at an increased risk for developing cancer, as is the liver (which should not be a surprise given how it is exposed to acetaldehyde). Finally, compared to women who do not drink, women who have at least three drinks per week have a 15% greater risk of breast cancer.
11.5.2. Wernicke-Korsakoff Syndrome
One of the common side effects of alcoholism is vitamin deficiency. People who drink heavily often have nutritional deficits on top of the effects of ethanol. A particularly common deficit is thiamine (vitamin B1) deficiency. This can be caused by malnutrition, decreased absorption of thiamine by the gastrointestinal tract, a reduction in stored thiamine due to liver disease, or a combination of these factors.
Wernicke Korsakoff Syndrome [3:49]
Thiamine deficiency is linked to two separate but related neurological disorders. The earlier phase of the syndrome is Wernicke encephalopathy (encephalopathy literally means “disease of the brain”). There are three characteristic symptoms associated with Wernicke encephalopathy: confusion, ophthalmoplegia, and ataxia. Ophthalmoplegia refers to paralysis of eye muscles (the -plegia suffix is also used in paraplegic). Ataxia is a general lack of motor control. These symptoms are the result of degenerative changes to the thalamus and hypothalamus. If thiamine replacement is initiated soon after diagnosis, the signs of Wernicke encephalopathy can be resolved.
The second disorder is Korsakoff syndrome, sometimes referred to as Korsakoff psychosis. Korsakoff is more slowly developing and longer-lasting but is still a consequence of thiamine deficiency. The main symptoms are amnesia (both retrograde and anterograde) and confabulation, or distorted memories that the patient is confident in despite being bizarre or false. As you would expect, these symptoms are caused by damage to the hippocampus and other areas of the brain associated with memory. Symptoms of Korsakoff generally appear as signs of Wernicke start to subside. Unless Wernicke is treated promptly and appropriately, Korsakoff becomes permanent and often irreversible.
Although both conditions are related to thiamine deficiency, it is possible to have one and not the other. Wernicke encephalopathy can occur in people without a history of alcohol use, and people can develop Korsakoff syndrome without having ever exhibited the former. Still, the two disorders do often overlap, in which cases they are simply referred to together as Wernicke-Korsakoff syndrome. Generally, the symptoms related to Wernicke encephalopathy will appear first, then subside as the symptoms of Korsakoff syndrome emerge. If left untreated, 75% of people suffering from Wernicke-Korsakoff syndrome end up with permanent brain damage, and about 20% die.
11.5.3. Fetal Alcohol Syndrome
Recall that in a previous section, it was noted that ethanol can cross the placenta. What happens if this occurs? In the worst case, assuming the fetus does not die, it may be born with a significant developmental disorder called fetal alcohol syndrome (FAS).
Drinking Alcohol During Pregnancy [7:04]
Fetal alcohol syndrome is part of a larger category of fetal alcohol spectrum disorders (FASD) and is the most severe disorder. It is estimated that 2–5% of school-aged children were born with FAS. FAS can be identified by distinct facial characteristics such as epicanthal folds, short palpebral fissures, flat midface and short nose, thin upper lip, micrognathia, indistinct philtrum, minor ear abnormalities, and low nasal bridge. Growth is delayed, resulting in reduced weight, height, and head circumference. There may be deformities in joints, limbs, and fingers as well as defects in the heart, kidneys, or bones.
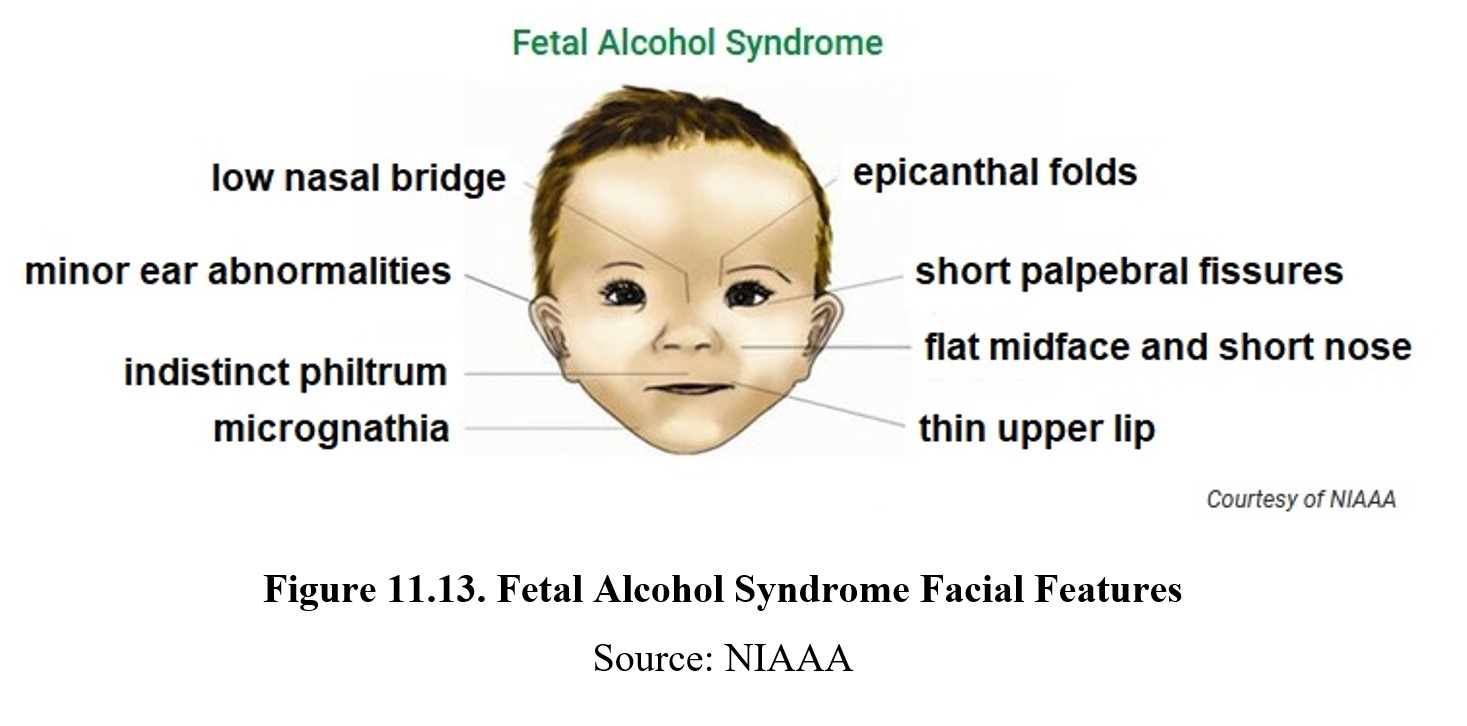
FAS results in neurological underdevelopment as well. People born with FAS may have difficulty with learning and memory and have a higher risk of psychiatric problems, especially ADHD and conduct disorders. These challenges may serve as significant obstacles to normal living. About 80% of individuals with FAS reported having difficulties with employment and independent living.
What causes FAS? A fetus has an undeveloped liver that is not properly equipped to metabolize ethanol. As a result, even very small concentrations of ethanol can build up and cause toxic effects. At the same time, the gestation period is essential for development, so even small amounts of toxins can cause lasting and irreparable harm. There is no safe level of alcohol consumption during pregnancy—abstinence is necessary to ensure normal development. As such, alcoholic beverages sold in the U.S. must contain a label warning that pregnant women should not drink alcoholic beverages due to the risk of birth defects.
11.6. Treatment of Alcohol Use Disorder
Section Learning Objectives
- Contrast alcohol abuse and dependence.
- State the DSM-5-TR criteria for alcohol use disorder.
- Discuss treatment options for alcohol use disorder.
According to the 2019 NSDUH, roughly 7.0% of males aged 12 and older and about 4.0% of females in the same age range had alcohol use disorder (AUD). Over 95,000 fatalities in the U.S. each year are attributed to excessive use of alcohol (CDC, 2016). In 2019, alcohol impairment accounted for 28.0% of driving fatalities (NCSA, 2019). Alcohol misuse and abuse carry a hefty economic burden on the country.
11.6.1. Alcohol Abuse vs. Alcohol Dependence
Though many people believe these to be one and the same, there is a fine line of distinction between alcohol abuse and alcohol dependence. Alcohol abuse is less severe than dependence. An alcohol abuser is one who chooses to consume alcohol and when to do so. An alcohol abuser can typically maintain some degree of awareness when drinking excessive alcohol. A person with alcohol dependence experiences cravings and has no control over their drinking. The person will compulsively seek out alcohol and experience withdrawal if they are absent from alcohol.
11.6.2. DSM-V Criteria for Alcohol Use Disorder
DSM-V lists eleven criteria for diagnosis of alcohol use disorder (AUD). Meeting any two criteria within a year’s time forms a positive diagnosis for AUD. The severity of AUD is dependent on the number of criteria that are met: two to three, mild substance use disorder; four to five, moderate substance use disorder; six or more, severe substance use disorder. What is an alcohol use disorder?
11.6.3. Treatment of AUD
The single most important predictor of outcome in recovery from alcohol use disorder (AUD) is the duration in which the client remains engaged in therapy. An alcohol treatment program generally consists of psychotherapy combined with pharmacotherapy. As is the case with mental disorders, this combination of approaches is more effective than either approach alone.
Another frequent approach is Alcoholics Anonymous (AA), which is a 12-Step program using self-improvement and empowerment and belief in one’s ability to replace unhealthy behaviors with healthier choices. Founded in 1935, AA is an outpatient self-help program that provides group treatment with peers who are also looking to cure their AUD plus a sponsor who is recovering from alcohol problems and committed to helping others stay sober.
Pharmacotherapy includes three drugs that have been approved by the FDA for the treatment of alcohol use disorder.
Drug Treatments for Alcohol Abuse [2:35]
Disulfiram (Antabuse®) was approved by the FDA in 1951 as a treatment of alcohol use disorder. It inhibits the enzyme acetaldehyde dehydrogenase (ALDH), which converts acetaldehyde to acetic acid and water. It is considered an alcohol deterrent used for aversion therapy or negative reinforcement, wherein the user is “punished” for consuming alcohol. Blocking ALDH causes an accumulation of acetaldehyde in the body resulting in the very unpleasant mal rouge syndrome.
Acamprosate (Campral®) was approved by the FDA in 2004. It is hypothesized to stabilize the imbalance between inhibitory GABA and excitatory glutamate systems in the brain. When used to treat alcoholics, acamprosate reduces cravings for alcohol and increases days of abstinence from alcohol. It also reduces relapse in alcoholics who have stopped drinking. Acamprosate makes it easier to maintain abstinence.
The opioid antagonist naltrexone was approved by the FDA in 1994 for the treatment of alcohol dependence. Administered following clearing alcohol from the body, naltrexone prevents experiencing pleasure upon drinking ethanol. Recall that there is an opioid involvement in alcohol-induced reward and reinforcement. Blocking opioid receptors restores GABA inhibition of dopamine release. Since ethanol is no longer rewarding, the subject is less likely to continue drinking. But naltrexone does not reduce craving for alcohol nor does it mitigate alcohol withdrawal. It is most effective when combined with other treatments.
A daily oral form of naltrexone (Revia®) is available as is depo-naltrexone (Vivitrol®), a once-monthly extended-release injectable form of naltrexone. Currently in clinical trials is a long-term naltrexone implant that slowly dissolves and continuously released the naltrexone dose for about six months.
Chapter Summary and Review
In this chapter, we took a deep dive into alcohol, also known as ethanol. We described its history, legislation, prevalence, and how it is measured. We learned about the pharmacokinetics of alcohol, starting with its absorption and distribution before exploring factors that influence its metabolism. We examined its mechanisms of action, effects in the CNS and elsewhere, and certain health risks associated with its use.
That is all for this chapter and this unit. There was a lot of information in this chapter, but, considering the extensive use of alcohol by the population, it is probably worth it to go over this information in detail. Make sure to take your time to study it properly. Best of luck, and do not hesitate to reach out to your instructor if you have any questions.
Chapter 11 Practice Questions
Answer the following questions:
- Do adults engage in riskier drinking behaviors than adolescents?
- If an alcoholic beverage is 90-proof, what is its percent ABV?
- On average, how many standard drinks will put you over the legal driving limit?
- Name three reasons why women tend to have higher BACs from the same amount of alcohol compared to men.
- At what BAC does mild motor impairment begin?
- Jamie is out drinking and says that he is seeing double. What is his BAC likely at?
- What percentage of ethanol is metabolized in the body?
- Which enzyme polymorphism metabolizes acetaldehyde at a near-inactive rate?
- What does disulfiram do, and how does it work?
- Annette says that alcohol always follows zero-order kinetics. How would you respond to her?
- What are the three types of tolerance to alcohol that one can develop?
- Explain how the ingestion of ethanol ends up increasing dopamine levels in the nucleus accumbens.
- What are the stages of liver disease?
- Name five parts of the body that see an increased risk of cancer due to alcohol use.
- What are the symptoms of Wernicke-Korsakoff syndrome? Be sure to distinguish between the two disorders.
- Is it possible to safely drink alcohol during pregnancy? Discuss fetal alcohol syndrome.
- What drugs are FDA-approved for the treatment of alcohol use disorder and how do they work?
2nd edition
